The second ionization energies of some Period 4 elements are Identify the orbital from which ionization occurs
Question:
The second ionization energies of some Period 4 elements are
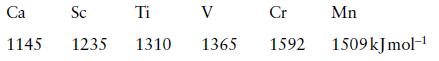
Identify the orbital from which ionization occurs and account for the trend in values.
Transcribed Image Text:
Ca Sc 1145 1235 Ti 1310 V 1365 Cr 1592 Mn 1509 kJmol-¹
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To identify the orbital from which ionization occurs for each of the Period 4 elements and account for the trend in values we need to understand the e...View the full answer

Answered By

Surendar Kumaradevan
I have worked with both teachers and students to offer specialized help with everything from grammar and vocabulary to challenging problem-solving in a range of academic disciplines. For each student's specific needs, I can offer explanations, examples, and practice tasks that will help them better understand complex ideas and develop their skills.
I employ a range of techniques and resources in my engaged, interesting tutoring sessions to keep students motivated and on task. I have the tools necessary to offer students the support and direction they require in order to achieve, whether they need assistance with their homework, test preparation, or simply want to hone their skills in a particular subject area.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
The first and second ionization energies of K are 419 kJ/mol and 3052 kJ/mol, and those of Ca are 590 kJ/mol and 1145 kJ/mol, respectively. Compare their values and comment on the differences.
-
In the paper Ionization energies of atoms and atomic ions (P.F. Lang and B.C. Smith, J. Chem. Educ., 2003, 80, 938) the authors discuss the apparent irregularities in the first and second ionization...
-
Without referring to your text, predict the trend of second ionization energies for the elements sodium through argon. Compare your answer with the data in Table 12.6. Explain any differences. Table...
-
All of the following represent trends shaping HR over the past decade, according to our reading, except: Organizations are spending more on the expansion of physical office space Employees are...
-
Given the following information for Lightning Power Co., find the WACC. Assume the companys tax rate is 35 percent. Debt: 8,000 6.5 percent coupon bonds outstanding, $1,000 par value, 25 years to...
-
Which should be the stronger base: ammonia, NH 3 , or nitrogen trifluoride, NF 3 ? F- N-F H - N-H Ammonia Nitrogen trifluoride
-
Consider two alternatives, each of which will accomplish the same EPA-mandated pollution control. Using an incremental IRR analysis, determine the preferred alternative assuming MARR is 20...
-
Baucom Manufacturing Corporation was started with the issuance of common stock for $50,000. It purchased $7,000 of raw materials and worked on three job orders during 2012 for which data follow....
-
x- 70 mm y 20 mm Eample 6: Determine the moment of inertia for the following area about its x centroidal axis. 40 mm 30 mm. X 25
-
What are the values of the n, l, and m l quantum numbers that describe the 5f orbitals?
-
How many orbitals are there in a shell of principal quantum number n?
-
Fruit flies flight dynamics are interesting to study because they provide a proof-of-concept framework and inspiration for the invention of man-made machines. In an experiment (Roth, 2012), flies are...
-
The income statement has been prepared by Daisy Ltd's accountant for the current year ending December 31, 2022 is as follow: Sales 8,000,000 Cost of goods sold 5,400,000 ...
-
A firm is currently partially financed with zero-coupon debt that promises to repay bondholders $200 at maturity. These bonds mature one year from today at t-1. The firm is in a very risky industry,...
-
The Mexican inflation rate is 3.11 per year. One year ago the spot exchange rate was 0.22 BRL (Brazilian Reals) per MXN (Mexican Pesos). Today it is 0.27 BRL (Brazilian Reals) per MXN (Mexican...
-
CSU, Inc., is a calendar year S corporation. CSU's Form 1120S shows nonseparately stated ordinary income of $120,000 for the year. Taewon owns 30% of the CSU stock throughout the year. The following...
-
At the 1 January 20X1 the balance on Alex's plant and equipment cost account was 67,400 and accumulated depreciation was 13,400. Plant and equipment are depreciated at 12.5% per annum using the...
-
Use BONDVAL to find the YTM of the following $1,000 par value bonds. 2 Market Price Coupon rate Term $752.57 6.590 15.5yrs $1,067.92A S915.05 12.590 2.5yrs 7.24% 8.5yrs
-
What are some of the various ways to implement an awareness program?
-
By considering the effect of each symmetry operation of the D 3h point group on the symmetric deformation mode shown in Fig. 3.14, confirm that this mode has A 2 '' symmetry. Figure 3.14 Symmetric...
-
Assign a point group to each member in the series (a) CCl 4 , (b) CCl 3 F, (c) CCl 2 F 2 , (d) CClF 3 (e) CF 4 .
-
The point group of BrF 3 is C 2v . Draw the structure of BrF 3 and compare your answer with the predictions of the VSEPR model.
-
In your job as a mechanical engineer you are designing a flywheel and clutch-plate system like the one in (Figure 1). Disk A is made of a lighter material than disk B, and the moment of inertia of...
-
13-19. Determine the maximum force P that can be applied to the handle so that the A-36 steel control rod BC does not buckle. The rod has a diameter of 25 mm. T 350 mm P 250 mm 800 mm Prob. 13-19
-
1. Determine the angular velocity of link OB if the piston has a velocity of 2 m/s to the right at the instant shown. A VA = 2 m/s 180 mm B 120 mm O 160 mm

Study smarter with the SolutionInn App


