Use the following experimental data to determine E o Cu 2+ /Cu , and comment on the
Question:
Use the following experimental data to determine EoCu2+/Cu, and comment on the need (or not) to make use of all the data given.
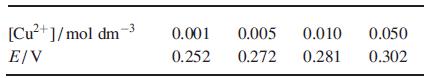
Transcribed Image Text:
[Cu²+]/mol dm E/V 0.001 0.005 0.010 0.252 0.272 0.281 0.050 0.302
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
To determine EoCu2Cu standard reduction potential of Cu2Cu we can use the Nernst equation which relates the standard reduction potential Eo to the mea...View the full answer

Answered By

Abdul Wahab Qaiser
Before working at Mariakani, I volunteered at a local community center, where I tutored students from diverse backgrounds. I helped them improve their academic performance and develop self-esteem and confidence. I used creative teaching methods, such as role-playing and group discussions, to make the learning experience more engaging and enjoyable.
In addition, I have conducted workshops and training sessions for educators and mental health professionals on various topics related to counseling and psychology. I have presented research papers at conferences and published articles in academic journals.
Overall, I am passionate about sharing my knowledge and helping others achieve their goals. I believe that tutoring is an excellent way to make a positive impact on people's lives, and I am committed to providing high-quality, personalized instruction to my students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Analytics mindset Final Project 4 Gamification Part 1: Background You are the chief technology officer (CTO) of an international bank. A key component of your job is to manage risk within the bank...
-
Write a literature review for your study. See below for an example of a literature review. Your literature review should provide both analysis and synthesis of previous studies as related to the...
-
The cylinder in the figure has a radius of 2 ft and rolls without slipping on the horizontal surface. The bar AB is attached to the periphery of the cylinder by a pin at point A. The other end of the...
-
Name and describe three drivers for counterattacks.
-
What are the functions or responsibilities of the policy committee and executive subcommittee in a multi office firm?
-
At 3:00 a.m. on November 22, 2010, 16-year-old Sydney McLemore was driving a Mazda3 with her friend, Natalie Hurst, in the front passenger seat. The vehicle was traveling south on Ross Bridge Highway...
-
The following events took place for Air Temp Manufacturing Company during January, the first month of its operations as a producer of digital thermometers: a. Purchased $68,000 of materials. b. Used...
-
Discuss at least five of the formatting decisions you would have to make when typing a table. Compare the decisions you provided to those of two of your classmates. Do you agree or disagree with the...
-
Consider the half-reaction: If the ratio of concentrations of [MnO 4 ] : Mn 2+ is 100:1, determine E at pH values of (a) 0.5; (b) 2.0; (c) 3.5 (T = 298 K). Over this pH range, how does the ability...
-
Given that K sp for AgI is 8:51 10 17 , and E o Ag + /Ag = +0:80 V, calculate E o for the reduction step: Hence confirm the statement in Section 8.3 that reduction of silver(I) when in the form of...
-
Mildred Enterprises provides the following information relative to its defined benefit pension plan. Balances or Values at December 31, 2010 Defined benefit...
-
Maryland Light, a U.S. manufacturer of light fixtures, is considering an investment in Japan. The dollar cost of equity for Maryland Light is 14.92%. You are in the corporate treasury department, and...
-
The dollar cost of debt for Healy Consulting, a U.S. research firm, is 7.96%. The firm faces a tax rate of 40% on all income, no matter where it is earned. Managers in the firm need to know its yen...
-
Your start-up company has negotiated a contract to provide a database installation for a manufacturing company in Poland. That firm has agreed to pay you $182,500 in three months when the...
-
Suppose the interest on Russian government bonds is 7.9%, and the current exchange rate is 26.1 rubles per dollar. If the forward exchange rate is 26.6 rubles per dollar, and the current U.S....
-
Which of the following sources of new earnings volatility represents the effect of business versus financial risk (discuss the rationale for your decisions): a. Pajala AB in Sweden is considering...
-
Versa Tools Inc. is a small tool and die manufacturing shop located in southwestern Ontario. The companys main shareholder, Arthur Eshelman, is becoming increasingly concerned about the safety and...
-
During 2012, Cheng Book Store paid $483,000 for land and built a store in Georgetown. Prior to construction, the city of Georgetown charged Cheng $1,300 for a building permit, which Cheng paid. Cheng...
-
Suggest a synthesis of neptunocene from NpCl 4 .
-
Explain why stable and readily isolable carbonyl complexes are unknown for the lanthanoids.
-
The elements Eu and Gd display certain similarities with Mn and Fe. Discuss this statement.
-
The income statement reports earned income on _____, and the cashflow statement is based on ______. Question 4Answer a. A cash-basis, cash-basis accounting b. A cash-basis, accrual-basis accounting...
-
Halloween candy prices are estimated to be 14% higher this year. Kroger decided to run a price promotion for Halloween candy beginning two weeks before the 31st. They know the elasticity of demand...
-
Electing Offices for a Club: The following members are being voted into office: N = {Alan, Bill, Cathy, David, Evelyn} Assuming they are all eligible to be elected and can only hold one office, list...

Study smarter with the SolutionInn App


