Use the Kapustinskii equation and the ionic and thermochemical radii given in Resource section 1 and Table
Question:
Use the Kapustinskii equation and the ionic and thermochemical radii given in Resource section 1 and Table 4.10, and r(Bk4+) = 96 pm to calculate lattice enthalpies of
(a) BkO2,
(b) K2SiF6,
(c) LiClO4.
Table 14.10.
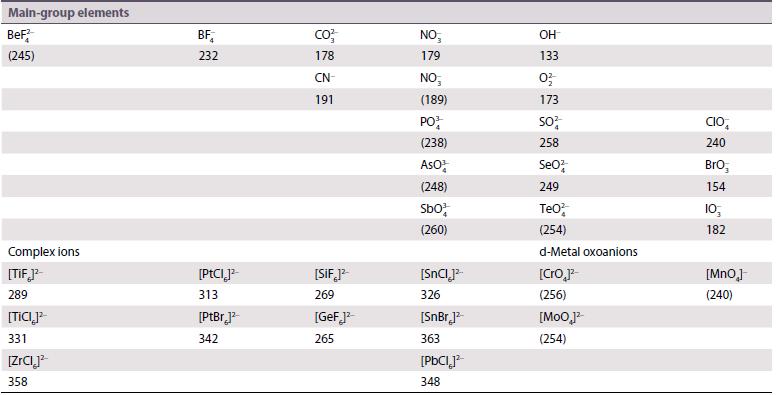
Transcribed Image Text:
Main-group elements BeF² (245) Complex ions [Tif]²- 289 [TICI]² 331 [ZrCI² 358 BF 232 [PtCl]²- 313 [PtBr]² 342 CO²/ 178 CN- 191 [SIF 1²- 269 [GeF]² 265 NO, 179 NO (189) PO (238) ASO¹ (248) Sb0% (260) ISnC] 326 [SnBr.]² 363 [PbCl]² 348 OH 133 02/ 173 S0² 258 SeO² 249 TeO² (254) d-Metal oxoanions [Cro 1²- (256) [MoO ]² (254) CIO 240 BrO₂ 154 10, 182 [MnO₂] (240)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The Kapustinskii equation is as follows Hlattice Z Z rm where Hlattice Lattice enthalpy in kJmol Pro...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
Use radius-ratio rules and the ionic radii given in Resource section 1 to predict structures of (a) UO 2 , (b) FrI, (c) BeS, (d) InP.
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The table below lists the ionic radii for the cations and anions in three different ionic compounds. Each compound has either the NaCl, CsCl, or ZnS type cubic structure. Predict the type of...
-
Provide an detailed overview on the topic indirect pay/benefits. Use the information given: Indirect Pay: any type of employer-provided reward (or "benefit") that serves an employee need but is not...
-
You have just arranged for a $1,800,000 mortgage to finance the purchase of a large tract of land. The mortgage has a 7.8 percent APR, and it calls for monthly payments over the next 30 years....
-
Statistically speaking, what is the most dangerous job in the United States?
-
Jason Cannon purchased a defective tractor for which Cannon brought numerous causes of action against Bodensteiner Implement Company (Bodensteiner). One of the claims was breach of express...
-
Elvira Industries had the following transactions. 1. Borrowed $5,000 from the bank by signing a note. 2. Paid $2,500 cash for a computer. 3. Purchased $450 of supplies on account. Instructions (a)...
-
c) Show that 2x+1 x (x+1) 1-2x2 = 2x + Hence, evaluate xx+1) S 2x+1 dx by first expressing the integrand as sum of partial fractions.
-
Determine the first four terms in the expression for the Madelung constant calculation for the CsCl structure.
-
On the basis of the factors that contribute to lattice enthalpies, place LiF, SrO, RbCl, AlP, NiO, and CsI, all of which adopt the rock-salt structure, in order of increasing lattice enthalpy.
-
Repeat Problem 4 for a school with 350 students. Data from in problem 4 (a) In a school of 150 students, one of the students has the flu initially. What is In? What is So? (b) Use these values of Io...
-
Mary is 32 and wants to buy her first home .A graduate of the Algonquin school of business - she loved the Investment Planning course J Has $80,000 in RRSPs (RBC Bond Fund), $70,000 in her TFSA (all...
-
A lens with f= +15 cm is in contact with a lens with f = -20 cm. Part A What is the effective focal length of the combination? Express your answer with the appropriate units. feff = Value Submit...
-
If $10,000 is invested in a certain business at the start of the year, the investor will receive $3,000 at the end of each of the next four years. What is the present value of this business...
-
What strategic initiatives might a retailer have implemented to address the challenges stemming from the COVID-19 pandemic, ensuring business continuity and resilience in the face of unprecedented...
-
Von Bora Corporation is expected pay a dividend of $2.17 per share at the end of this year and a $2.42 per share at the end of the second year. Immediately after Von Bora pays the $2.42 per share...
-
Information on a packet of seeds claims that the germination rate is 92 %. Whats the probability that more than 95% of the 160 seeds in the packet will germinate? Be sure to discuss your assumptions...
-
In the synthesis of the keto acid just given, the dicarboxylic acid decarboxylates in a specific way; it gives Explain. HO rather than HO
-
Four pK a values (1.0, 2.0, 7.0, 9.0) are tabulated for the acid H 4 P 2 O 7 . Write equations to show the dissociation steps in aqueous solution and assign, with reasoning, a pK a value to each step.
-
The structure of H 5 DTPA (see Box 4.3) is shown below: (a) Write equilibria to show the stepwise acid dissociation of H 5 DTPA. Which step do you expect to have the largest value of K a ? (b) In the...
-
In aqueous solution, boric acid behaves as a weak acid (pK a = 9:1) and the following equilibrium is established: (a) Draw the structures of B(OH) 3 and [B(OH) 4 ]. (b) How would you classify the...
-
A 4-DOF robot arm was shown in the figure 1. For this robot: a) Assign coordinate frames based on the D-H representation. b) Fill out the D-H parameters table. c) Find the link transformation matrix...
-
2. Compare a Magnetic Drive Sealless Pump with a Canned Pump. a. Discuss one common feature of both pumps [1m] b. Discuss three differences between these 2 pump types [3m]
-
Page 2 2. In designing structures and machines, it is important to consider strength of material so that the material should have an adequate strength to resist applied loads or forces and retain its...

Study smarter with the SolutionInn App


