Using electronegativity values of 1.57 for Be and 0.79 for Cs and a Ketelaar triangle (Fig. 2.28),
Question:
Using electronegativity values of 1.57 for Be and 0.79 for Cs and a Ketelaar triangle (Fig. 2.28), predict what type of compound might form between these elements.
Figure 2.28.
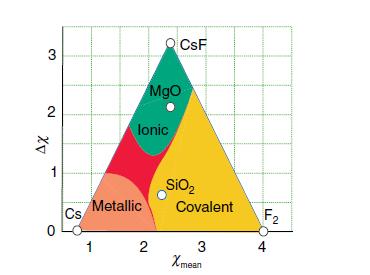
Transcribed Image Text:
3 2 ΔΧ 1 0 Cs Metallic 1 lonic MgO CsF 2 SiO₂ Covalent 3 Xmean F₂ 4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 20% (5 reviews)
To predict the type of compound that might form between beryllium Be and cesium Cs we can use the el...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
Use the Ketelaar triangle in Fig. 2.28 and the electronegativity values in Table 1.7 to predict what type of bonding is likely to dominate in (a) BCl 3 , (b) KCl, (c) BeO. Figure 2.28. Table 1.7. 3 2...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
A five-year follow-up study was carried out in a certain metropolitan area to assess the relationship of diet and weight to the incidence of stomach cancer. Data were obtained on n = 2,000 subjects....
-
Consider two farmers, A and B, produce farm products and sell in the same market. Assume that the supply of the two farmers products are the same but the demand for Farmer Bs product is relatively...
-
At a professional conference just a few days ago, Darcy Kramer, the president of Luard Corporation, learned how the concept of controllability relates to performance evaluation. In preparing to put...
-
How might the data reduction approach be used in auditing?
-
Respondents Varsity Brands, Inc., Varsity Spirit Corporation, and Varsity Spirit Fashions & Supplies, Inc., design, make, and sell cheerleading uniforms. Respondents have obtained or acquired more...
-
FIFO and LIFOPeriodic and Perpetual Inventory information for Part 311 of Seminole Corp. discloses the following information for the month of June. June 1 Balance 300 units @ $10 June 10 Sold 200...
-
A 250 kg beam is raised through 25 m at a constant velocity by a crane. a) Draw an FBD for the beam. b) Determine the work done by the crane on the beam. c) Determine the work done by gravity on the...
-
Which salts of francium would be the least soluble? How could francium be precipitated and isolated from solution that also contains sodium ions?
-
The molecular geometries of crown ether derivatives play an important role in capturing and transporting alkali metal ions. K. Okano and co-workers (see K. Okano, H. Tsukube, and K. Hori,...
-
Refer to the research study concerning the effect of social factors on reading and math scores in Section 3.8. We justified studying just the reading scores because there was a strong correlation...
-
It has been said that Hart's theory of law incorporates elements of both Natural Law Legal Theory and Legal Realism. Discuss.
-
In finance, what does the word "maturity" mean? Explain.
-
Why does changing the speed of sound change the resonant frequencies of the vocal tract? (think ALL the way back to Basic Acoustics and consider how the speed of sound changes wavelength).
-
Is SAP EWM categorized as business intelligence software or transactional applications, delineating its functional role within enterprise resource planning (ERP) systems and warehouse management...
-
Classify the following medicines. Write OTC if they are Over-the-Counter- Drugs and Rx if they are Prescription Medicines. 1. Codeine 2. Fentanyl 3. Kremil-S 4. Morphine 5. Bonamine 6. Penicillin 7....
-
Zinc and magnesium metal each react with hydrochloric acid to make chloride salts of the respective metals and hydrogen gas. A 10.00- g mixture of zinc and magnesium produces 0.5171 g of hydrogen gas...
-
Find i 0 (t) for t > 0 in the circuit in Fig. 16.72 . 2 + Vo 1 7.5e-2t u(t) V ( +) 4.5[1 u(t)]V 0.5v. 1H
-
Does VB theory indicate that the diatomic molecule He 2 is a viable species? Rationalize your answer.
-
(a) Draw resonance structures for CO, choosing only those that you think contribute significantly to the bonding. (b) Figure 2.15a shows an MO diagram for CO. Two MOs are illustrated by schematic...
-
Using VB theory and the Lewis structure model, determine the bond order in (a) H 2 , (b) Na 2 , (c) S 2 , (d) N 2 (e) Cl 2 . Is there any ambiguity with finding the bond orders by this method?
-
1. Download the historical data series on economic growth for the past three decades for the Canadian and US economy (from Q3 1993 to Q2 2023). 1. Graph the economic growth and determine the...
-
Audit Plan - Located under the Written Assignments tab Review the template and the Assignment Rubric for specifics to include in the plan. Use the Audit Plan Template to develop an internal coding...
-
The Role of Financial Statements in Business Decision Making [WLOs: 1, 2, 3] [CLOs: 1, 2] Prior to beginning work on this discussion, please read the articles: Researching Public Companies Through...

Study smarter with the SolutionInn App


