Calculate the enthalpy of mixing for HCl from the enthalpy of solution data reported in Table 6.1.
Question:
Calculate the enthalpy of mixing for HCl from the enthalpy of solution data reported in Table 6.1.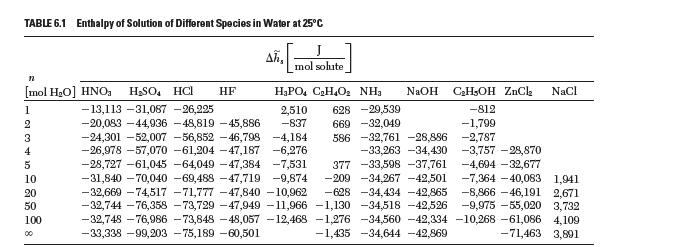
Transcribed Image Text:
TABLE 6.1 Enthalpy of Solution of Different Species in Water at 25C Ah, J mol solute n NaOH CH5OH ZnCl NaCl HPO4 CH4O NH 2,510 628 -29,539 -812 669 -32,049 -1,799 -2,787 586 -32,761 -28,886 -33,263 -34,430 [mol HO] HNO HSO4 HC -13,113 -31,087 -26,225 -20,083-44,936 -48,819 -45,886 -837 -24,301 -52,007 -56,852 -46,798 -4,184 -26,978 -57,070 -61,204 -47,187 -6,276 -3,757 -28,870 -28,727 -61,045 -64,049 -47,384 -7,531 377 -33,598 -37,761 -4,694 -32,677 -31,840 -70,040 -69,488 -47,719 -9,874 -209 -34,267 -42,501 -7,364-40,083 1,941 -32,669 -74,517 -71,777 -47,840 -10,962 -628 -34,434 -42,865 -8,866-46,191 2,671 -32,744 -76,358 -73,729 -47,949 -11,966 -1,130 -34,518 -42,526 -9,975 -55,020 3,732 -32,748 -76,986 -73,848 -48,057 -12,468 -1,276 -34,560 -42,334 -10,268 -61,086 4,109 -33,338 -99,203 -75,189 -60,501 -1,435 -34,644 -42,869 -71,463 3,801 1 2 3 4 5 10 20 50 100 00 HF
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Shehar bano
I have collective experience of more than 7 years in education. my area of specialization includes economics, business, marketing and accounting. During my study period I remained engaged with a business school as a visiting faculty member and did a lot of business research. I am also tutoring and mentoring number of international students and professionals online for the last 7 years.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
In a process at your plant, you are mixing two liquids: benzene (1) and 2-propanol (2). You would like to create a molar enthalpy vs. composition diagram for this mixture and have found molar...
-
Use the data of Table 13.1 to calculate the enthalpy of solution of LiCl. Data given in Table 13.1 TABLE 13.1 Data for Calculating Enthalpy of Solution Compound LiF NaF KF RbF LICI NaCl KCI RbCl NaOH...
-
An experiment is performed to measure the enthalpy of mixing of chloroform, CHCl3, and acetone, C3H6O. In this experiment, pure species inlet streams of different compositions are mixed together in...
-
Contact local employers and ask for copies of their employee handbooks. If none are available, research parent companies of local employers online to see if their employee handbooks are available....
-
Based on the case : Polaris & Victory: Entering and Growing the Motorcycle Business Explain: 1- External Environment 2- General Environment 3- Menneto knew his bikes were good, but had they been...
-
A tenured professor falls down the stairs at her university and suffers a brain injury and is unable to work. She is told by the university that in order to be in compliance, she must apply for FMLA...
-
In the Young's interference experiment shown in Fig. 5-5-9pp, the normalized power spectral density \(\widehat{\mathcal{G}}(v)\) of the light is measured at point \(Q\) by a spectrometer. The mutual...
-
Gasoline is sold through local gasoline stations under perfectly competitive conditions. All gasoline station owners face the same long-run average cost curve given by AC = .01q 1 + 100/q and the...
-
Apex Limited (APL) is a management consultancy company headquartered in Singapore. After the successful implementation of a share option plan for the senior management team in 20x1, the company has...
-
What is the heat requirement to dilute an inlet aqueous stream of 50% NaOH, by weight, to a fi nal concentration of 10%?
-
How does the enthalpy of mixing data for H2SO4 given by Equation (6.24) compare to the enthalpy of solution data from Table 6.1? Is the agreement reasonable? What are the reasons they may be...
-
In testing for co integration between gfr and pe in Example 18.5, add t2 to equation (18.32) to obtain the OLS residuals. Include one lag in the augmented DF test. The 5% critical value for the test...
-
George bought Blackacre for $500,000. George put $50,000 in cash and the rest of the purchase is financed by a $450,000 nonrecourse loan. George claims $100,000 in depreciation deductions, 7 years...
-
ABC manufacturing has produced parts that can only be sold to a single buyer, the buyer has refused to make additional payment for the remaining goods. What exception can ABC manufacturing seek help...
-
what ways can organizational leaders employ sophisticated time management frameworks to cultivate a culture of accountability and excellence, fostering optimal performance across multifaceted teams...
-
The manager of a home appliancecompany, where welding is a very common operation, is concerned that too much pollution in the workplace may be negatively affecting employee morale. The manager...
-
16. The data referred to in this question were collected on 41 employees of a large company. The company is trying to predict the current salary of its employees from their starting salary (both...
-
How are persuasive requests and sales letters similar and how are they different?
-
Should we separate the debt and equity features of convertible debt? Team 1: Pro separation: Present arguments in favor of separating the debt and equity features of convertible debt. Team 2: Against...
-
Assign the following molecules to point groups: (a) HF, (b) IF7 (pentagonal bipyramid), (c) XeO2F2, (see-saw), (d) Fe,(CO)9 (22), (e) Cubane, C8H8, (f) Tetrafluorocubane, C8H4F4 (23) cO Fe co 23
-
Which of the molecules in Exercises 12.9b and 12.10b can be? (a) Polar, (b) Chiral?
-
Consider the C3Yion NO;. Is there any orbital of the central N atom that can have a nonzero overlap with the combination 2pz (A) pz (B) pz (C) of the three O atoms (with z perpendicular to the...
-
400 mm web angle connection web angle connection 4000 mm 4000 mm In a residential building, a W310x52 section of ASTM A992 steel (Fy-345 MPa) is selected to support a point dead load of 20 kN at its...
-
Group building Roles that are directly related to the effectiveness of the organization are called?
-
We Supply Ltd is a small shop in Sasolburg. The following information for WeSupply Ltd is available: R Sales 300 000 Earnings after interest and tax 150 000 Preference dividends due 20 000 Preference...

Study smarter with the SolutionInn App


