The following data are available for ethylene at 24.95C. From these data, estimate the fugacity and the
Question:
The following data are available for ethylene at 24.95°C. From these data, estimate the fugacity and the fugacity coeffi cient of ethylene at 50.5 bar and 24.95°C.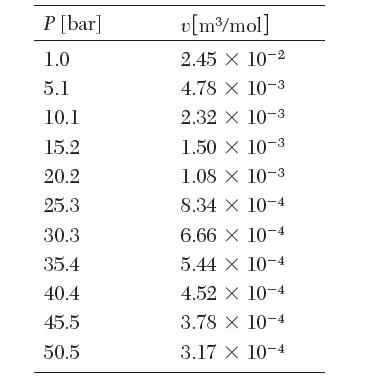
Transcribed Image Text:
P [bar] 1.0 5.1 10.1 15.2 20.2 25.3 30.3 35.4 40.4 45.5 50.5 v[m/mol] 2.45 10-2 4.78 X 10-3 2.32 X 10-3 1.50 X 10-3 1.08 x 10-3 8.34 X 10-4 6.66 X 10-4 5.44 x 10-4 4.52 X 10-4 3.78 x 10-4 3.17 x 10-4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Compute the CLV metric and then apply it in a simple breakeven analysis. The data below show the results of a Starbucks segmentation study. Each column shows the values for a typical customer in each...
-
The following data are from Gennings, Chinchilli, and Carter, (1989). An in vitro toxicity study of isolated hepatocyte suspensions was conducted to study the impact of combining carbon tetrachloride...
-
The velocity of water, v (m/s), discharged through a cylinder can be computed as f(H)= V2gH tanh ( 2gH 2L t V Where g = 9.81 m/s, H = initial head (m), L = pipe length (m), and t = elapsed time (s)....
-
Which of the following is part of Walmarts strategy regarding its supply chain? a) Acquiring e-commerce technology and expertise. b) Opening central warehouses. c) Changing how and when inventory is...
-
Determine two to three methods of using stocks and options to create a risk-free hedge portfolio. Support your answer with examples of these methods being used to create a risk-free hedge portfolio....
-
1. What factors did the court use to determine that the Local Fraternity may have assumed a duty here? Do you agree? Why or why not? 2. Why did the court find that Wabash College had no liability for...
-
A thin-walled cylindrical pressure vessel has mean diameter \(d=18\) in. and wall thickness \(h=0.25\) in. The vessel is made of filament-wound unidirectional composite material with all fibers...
-
Answer the following multiple-choice questions. 1. GAAP stands for: (a) Governmental auditing and accounting practices. (b) Generally accepted attest principles. (c) Government audit and attest...
-
Write the complete code to remove the node "banana" in the singly linked list shown in the Fig. 1. The sample code for the class Node is given below: public class Node { public String element; public...
-
Below is a plot of the natural log of the fugacity coeffi cient, ln (i) , of pure NH3 as a function of pressure at a temperature of 100C. From this plot, as best you can, determine the molar volume...
-
As accurately as you can, determine the fugacity of pure methane at 220 K and 69 bar.
-
Use the matrix solver in a program like Mathematica to solve (3.1.20) for a general k(vector) , and plot versus k, for k(vector) in two different directions in the plane. Determine the polarization...
-
Describe an example of how formal or informal mediation might be used to facilitate Ottos rights.
-
Describe how you would choose between personal values, organizational values, and your responsibility to the shareholders.
-
Describe how job analysis supports the creation of the job description?
-
Describe the key considerations in the design of products and services within the context of supply chain management and operations management.
-
Describe the process to establish base pay. Explain the incentives that companies use to attract domestic employees. Analyze the approach a company might use to determine base pay considering...
-
Will Starbucks ever return to the revenue and profit growth that it once enjoyed? Why or why not? By now, you should be familiar with the Starbucks story. After a trip to Italy in the early 1980s,...
-
Problem 2. (0.6 points, 0.2 points for each question) (a) A company turns its inventory 2 times a month. Its months-of-supply = Its days-of-supply = Please show your analysis below: _months. days. (1...
-
Given that p*(HzO) = 0.02308 atm and p (HzO) = 0.02239 atm in a solution in which 0.122 kg of a non-volatile solute (M = 241 g mol-1) is dissolved in 0.920 kg water at 293 K, calculate the activity...
-
Explain why Einstein's introduction of quantization accounted for the properties of heat capacities at low temperatures.
-
Account for the uncertainty relation between position and linear momentum in terms of the shape of the wave function.
-
Solve 0.44 * In (1+x) dx 0.44 In (1+x) dx (Type an integer or decimal rounded to three decimal places as needed)
-
Which school of thought believes that unions are necessary to counter corporate bargaining power?
-
1. Define mass media and explain why it is a socializing agent for children. 2. How is Obesity an issue for children's socialization? How do we address this issue as an early childhood educator? 3....

Study smarter with the SolutionInn App


