The following vaporliquid equilibrium data have been reported for a binary mixture of acetone (1) in water
Question:
The following vapor–liquid equilibrium data have been reported for a binary mixture of acetone (1) in water (2) at 1 atm. Test these data for thermodynamic consistency.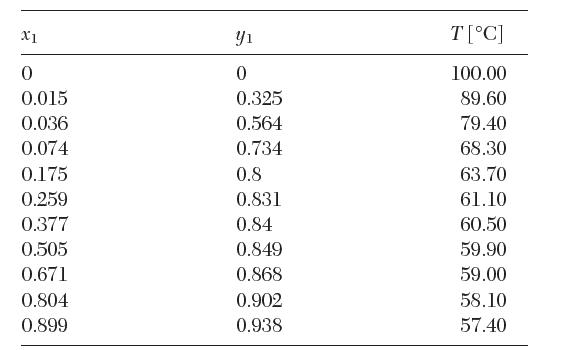
Transcribed Image Text:
X1 0 0.015 0.036 0.074 0.175 0.259 0.377 0.505 0.671 0.804 0.899 Y 0 0.325 0.564 0.734 0.8 0.831 0.84 0.849 0.868 0.902 0.938 T[C] 100.00 89.60 79.40 68.30 63.70 61.10 60.50 59.90 59.00 58.10 57.40
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)

Answered By

Asd fgh
sadasmdna,smdna,smdna,msdn,masdn,masnd,masnd,m asd.as,dmas,dma.,sd as.dmas.,dma.,s ma.,sdm.,as mda.,smd.,asmd.,asmd.,asmd.,asm
5.00+
1+ Reviews
15+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Use ThermoSolver to fi nd the activity coeffi cient model parameters for the data presented in Problems 8.55 and 8.56. Problems 8.55 The following vaporliquid equilibrium data have been reported for...
-
The following vaporliquid equilibrium data have been reported for a binary mixture of acetone (1) in chloroform (2) at 35. 17C. Test these data for thermodynamic consistency. X1 0 0.0821 0.1953...
-
A phase diagram for a binary mixture of ammonia (1) water (2) at 1 atm is shown in the following fi gure. Answer the following questions. Illustrate whenever possible the information you obtain from...
-
For each polynomial function, find (a) (-1), (b) (2), and (c) (0). f(x)=x5x4
-
After resolving their dispute, the two brothers encountered in Question 3-2 decide to resume their tax-accounting practice according to the terms of their original agreement. Again a dispute arises,...
-
Use analytical methods to evaluate the following limits. log2 x lim xo log3 x
-
Derive Equation 3.66. Uf 2vm + (3.66) E2 Vi+n2vm Et Em
-
The following gure shows the bill of material (BOM) for the Acme PolyBob, a product that has proven unsuccessful in capturing roadrunners. Complete the MRP records. All the information you need is...
-
CIVE 421: Reinforced Concrete Design Summer 2 2023 2nd Exam - Open Book Open Notes 1. Shear and moment design problem - continuous beam span As2 Section 2 Section 1 Wu As2 Asl L = 24 ft Only moment...
-
You wish to fi t the benzene (1)isooctane (2) system to the following model for gE: The system temperature of interest is 200C. After a literature search, the only vaporliquid equilibrium data at...
-
Test the liquidvapor equilibrium data for the binary system of methanol (1)water (2) at 40C presented in Problem 8.53 for thermodynamic consistency by using the area test. Problem 8.53 Liquidvapor...
-
Write balanced reactions for the destruction of S 2 O 2 -8 , Ag 2+, and H 2 O 2 by boiling.
-
Calculate the following: Suppose we deposit $10,000 in a savings account with interest compounded at 6% annually, what will be the money obtained at the end of 5 years? Suppose we take out a loan of...
-
1) The furnace is operated at 1600K. What are the most and least stable oxides at this temperature? 2) What is the maximum partial pressure of oxygen that allows reduction of CoO if the furnace is...
-
Consider the points P = (8,1), P = (-4,7), P3=(2, 6), P4 = (6,4), and P5 =(4,-9) of the plane. (i) For all i, j = 1, 2, ..., 5, find the distance di, between the points P and Pj. Show your work by...
-
today you borrowed 100,000 at 10% APR compounded monthly to buy a house. you will repay the mortgage with equal monthly payment over 10 years. First payment is due in a month. a) what is the...
-
In each of the following systems, a particle on a horizontal surface is acted on by two forces, F and F2. Find the i and j components of the resultant force, where j represents due north and i...
-
Perform an Internet search on social media monitoring to find companies that specialize in monitoring social media. Discuss two of these companies. Then find two more sites that allow free monitoring...
-
United Business Forms capital structure is as follows: Debt ............................................ 35% Preferred stock ........................... 15 Common equity .......................... 50...
-
Describe reasonable syntheses of Benzophenone, from each of the following starting materials and any necessary inorganic reagents. (a) Benzoyl chloride and benzene (b) Benzyl alcohol and bromobenzene...
-
The sex attractant of the female winter moth has been identified as the tetraene CH3(CH2)8CHCHCH2CHCHCH2CHCHCHCH2. Devise a synthesis of this material from 3, 6-hexadecadien-1-ol and allyl alcohol.
-
Hydrolysis of a compound a in dilute aqueous hydrochloric acid gave (along with methanol) a compound B, mp 164165C. Compound B had the molecular formula C16H16O4; it exhibited hydroxyl absorption in...
-
2. Look up one of the SBA guaranteed loans using the link below. Then write a paragraph describing the one you chose. www.sba.gov/content/sba-loans
-
The following tracer data were collected for a treatment reactor where 99% removal of a toxic pollutant is required. Given a first-order reaction relative to the pollutant concentration, what must be...
-
DoorDash is planning an IPO for the 4th quarter of 2020. The company is currently valued at $16B and plans to issue 16 million shares as part of its IPO at this valuation. These shares will have a...

Study smarter with the SolutionInn App


