The partial molar Gibbs energy of species 1 in a binary liquid mixture of species 1 and
Question:
The partial molar Gibbs energy of species 1 in a binary liquid mixture of species 1 and 2 is given by: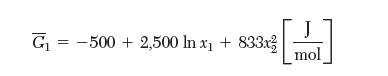

Transcribed Image Text:
G-500 + 2,500 In x + 833x J mol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)

Answered By

Romrick Albia
I am excellent student in my school. I have much interest in mathematics. I am studying from jawahar navodaya vidyalaya kaushambi and i am class topper at my school from 6 class to 12th class. I really like to clear the doubt of my classmates.
I have good skill of solving problems with easy way.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
A binary mixture of species a and b behaves as an ideal gas at 300 K and 1 bar. Calculate the partial molar Gibbs energy of species a, Ga, and the total solution Gibbs energy, g, at the following...
-
We have the following properties for a certain mixture for mixing at constant temperature and pressure: where S i , the pure-component molar entropy of component i, is given by Here S i , U i , and V...
-
At 25C and atmospheric pressure the volume change of mixing of binary liquid mixtures of species 1 and 2 is given by the equation V = x 1 x 2 (45x 1 +25x 2 ) where V is in cm 3 mol 1 . At these...
-
How does unemployment behave over the business cycle?
-
1- Describe the project manager's role in monitoring tasks. 2- Describe the project manager's role in directing resources. 3- Describe the project manager's role in managing change requests. 4-...
-
A tank is full of water. Find the work required to pump the water out of the spout. In Exercises 25 and 26 use the fact that water weighs 62.5 lb/ft 3 . 12 ft 6 ft 10 ft
-
Show that the standard deviation \(\sigma_{I}\) of the instantaneous intensity of partially polarized thermal light is \[ \sigma_{I}=\sqrt{\frac{1+\mathcal{P}^{2}}{2}} \bar{I} \]
-
Ajay is a rational, risk averse investor with $ 5,000 to invest for one year. He has decided to invest this amount in a high- technology firm and has narrowed his choice down to either AB Ltd. or XY...
-
1. Translate the following C code to RISC-V assembly (assume a, b, c are in x5, x6, x7): int a = 12; int b = 42; int c; c = b++ a; printf("%d", c);
-
The following expression describes the molar volume of a binary liquid mixture of species 1 and 2: Consider a mixture that contains 2 mol of species 1 and 4 mol of species 2. Answer the following...
-
You are running a manufacturing process where a mole fraction of 0.40 species 1 in a binary mixture of 1 and 2 is required at a fl ow rate of 5 mol/s and a temperature of 300 K. This is achieved by...
-
Multiply the following numbers by 10. a. 630 b. 4.6 c. 0.84 d. 0.065 e. 1.07
-
How do organizations strategically navigate the intricacies of stakeholder analysis and engagement to ensure buy-in and alignment during times of transformative change ?
-
What methodologies and frameworks, such as Kotter's 8-Step Process or Lewin's Change Management Model, offer nuanced insights into orchestrating successful organizational change initiatives?
-
How do leaders cultivate resilience and emotional intelligence within their teams, equipping individuals with the psychological tools necessary to navigate ambiguity and embrace change as a catalyst...
-
A seller believes he is pricing optimally. Her current unit margin rate is 15 percent. What must the actual price elasticity of demand equal if she is right?
-
How does the new nursing shortage impact the nursing shortage? Will it help in solving the issue of nursing shortage or add to it?
-
List effective tools for building interest in a persuasive request.
-
What will be the final value of DI after executing the following piece of code? Execute the instructions dependently one after another. CLD MOU CX,OFOH MOU AX.02874H MOU DI,01000H MOU ES, DI SUB...
-
An object of mass 2.0 g suspended from the end of a spring has a vibrational frequency of 3.0 Hz. Calculate the force constant of the spring.
-
Calculate the percentage difference in the fundamental vibration wave number of IH35Cl and 2H37CIon the assumption that their force constants are the same.
-
The wave number of the fundamental vibrational transition of 79BrB1Bris 323.2 cm-1, Calculate the force constant of the bond (mC9Br) = 78.9183 u, m(8IBr) = 80.9163 u).
-
Gain on Sale of Land* $4,000 MACRS Depreciation 7,500 Charitable Contributions 12,500 Sales 40,000 Interest Income 500 Cost of Goods Sold 32,000 Section 179 Expense 7,000 Tax-Exempt Income 2,000...
-
Atlee Inc. wants to determine the average collection period ratio for the last fiscal from the information provided: Accounts Receivables (Jan 01, 2020): $43,000 / Accounts Receivables (Dec 31,...
-
Critically explain under which circumstances it is not advisable for the investor not to sell down the fixed income instruments despite rising interest rate environment.

Study smarter with the SolutionInn App


