Question: Using data from Table 4.2, estimate the equilibrium bond length that would exist in a molecule of Xe2. TABLE 4.2 Lennard-Jones Parameters for Several Species
Using data from Table 4.2, estimate the equilibrium bond length that would exist in a molecule of Xe2.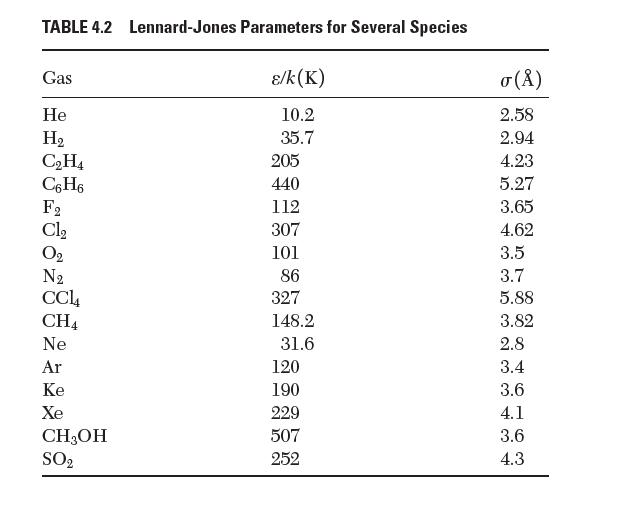
TABLE 4.2 Lennard-Jones Parameters for Several Species /k(K) 10.2 35.7 Gas He H CH4 C6H6 288 28 Cl CH4 Ne Ar Ke Xe CH3OH SO 205 440 112 307 101 86 327 148.2 31.6 120 190 229 507 252 () 2.58 2.94 4.23 5.27 3.65 4.62 3.5 3.7 5.88 3.82 2.8 3.4 3.6 4.1 3.6 4.3
Step by Step Solution
3.45 Rating (155 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


