Using the generalized compressibility charts, calculate the molar volume of ammonia at 92C and 306.5 bar. What
Question:
Using the generalized compressibility charts, calculate the molar volume of ammonia at 92°C and 306.5 bar. What phase is ammonia in?
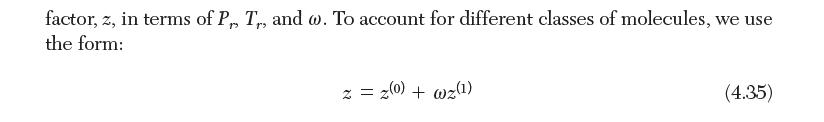

Transcribed Image Text:
GENERALIZED COMPRESSIBILITY CHARTS The principle of corresponding states invokes a unique generalized relation between the compressibility factor and reduced temperature and pressure for a given class of molecules. It is sometimes convenient to have graphs or tables that quantify this rela- tionship. In this section, we present charts and tabular data for the compressibility
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Madhvendra Pandey
Hi! I am Madhvendra, and I am your new friend ready to help you in the field of business, accounting, and finance. I am a College graduate in B.Com, and currently pursuing a Chartered Accountancy course (i.e equivalent to CPA in the USA). I have around 3 years of experience in the field of Financial Accounts, finance and, business studies, thereby looking forward to sharing those experiences in such a way that finds suitable solutions to your query.
Thus, please feel free to contact me regarding the same.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Ammonia burns in oxygen gas to form nitric oxide (NO) and water vapor. How many volumes of NO are obtained from one volume of ammonia at the same temperature and pressure?
-
Calculate the molar volume of saturated liquid and the molar volume of saturated vapor by the Redlich/Kwong equation for one of the following and compare results with values found by suitable...
-
Reconsider Prob. 384. Solve the problem using the generalized compressibility factor feature of the EES software. Again using EES, compare the specific volume of water for the three cases at 10 MPa...
-
Briefly explain the DHCP lease process. What packets are sent and when are they sent? (5 marks) Question 2 any three advantages of IPv6 over IPv4? How many classes are there in IPv4 and what is the...
-
Prepare ledger accounts for the transactions of Jane Gate's dental practice, listed in question A5.5. Refer to A5.5, Apr. 1 Jane Gate commenced her dental practice on 1 April by depositing 60,000 in...
-
The following transactions apply to Ozark Sales for Year 1: 1. The business was started when the company received $50,000 from the issue of common stock. 2. Purchased equipment inventory of $380,000...
-
Water flows through a 2-in.-diameter pipe with a velocity of \(15 \mathrm{ft} / \mathrm{s}\) as shown in Fig. P8.78. The relative roughness of the pipe is 0.004, and the loss coefficient for the exit...
-
Ryan Company wishes to prepare a forecasted income statement, balance sheet, and statement of cash flows for 2012. Ryans balance sheet and income statement for 2011 follow: Balance Sheet 2011 Cash ....
-
"How can an organization leverage automated backup verification processes to ensure the reliability and recoverability of backup data? Discuss the potential role of machine learning and artificial...
-
Use the RedlichKwong equation to calculate the size of vessel you would need to contain 30 kg of acetylene mixed with 50 kg of n-butane at 30 bar and 450 K. The binary interaction coeffi cient is...
-
Compare the compressibility factor of methane at Tr = 1.1 and Pr = 1.2 using the Peng Robinson equation of state and the compressibility charts. Repeat the calculations for methanol.
-
Solve Prob. 4.135 when h = 10.5 ft. PROBLEM 4.135 The 8-ft rod AB and the 6-ft rod BC are hinged at B and supported by cable DE and by ball-and socket joints at A and C. Knowing that h = 3 ft,...
-
You want to buy forward British sterling to be delivered eight months from today. The spot rate today is $1:49=. The rate of return in the United States is 3 percent per year; in Britain, 5 percent...
-
Explain the complexity of Algorithms ( 1. while(n>=0) { If(n>100) { for (i=1,i
-
What strategic management challenges are faced by international businesses that have business units scattered around the world? How can a business strategy help improve a firm's ability to compete?
-
Explain, with the help of some illustrative examples, the various foreign exchange risks. What are the ramifications, if the exchange rates are (i) fully covered, (ii) partly covered, and (iii) fully...
-
What proportion of customers rate the company with top box survey responses (which is defined as scale levels 4 and 5) on quality, ease of use, price, and service in the Customer Survey worksheet?...
-
The following information comes from the 2006 financial statements of McDonalds Corporation. Individual franchise arrangements generally include a lease and a license and provide for payment of...
-
3.16. For a system with non-identical service rates (see Sect. 3.5) and a limit of N jobs in the system (Eq. 3.13), obtain an expression for the mean service time per job, E[Ts], as a function of the...
-
Which one of the following compounds is most consistent with the infrared spectrum given in Figure 13.31? Explain your reasoning.
-
max for the * transition in ethylene is 170 nm. Is the HOMOLUMO energy difference in ethylene greater than or less than that of cis, trans-1,3-cyclooctadiene?
-
Which one of the C5H8 isomers shown has its max at the longest wavelength?
-
You observe that the inflation rate in the United States is 1.5 percent per year and that T-bills currently yield 2.0 percent annually. Use the approximate international Fisher effect to answer the...
-
The accompanying table provides the inflation rate in the year 2005 and the average inflation rate over the period 20062019 for eight different countries. Country Inflation rate in 2005 Average...
-
Q1. From the following balances prepare Trading and Profit & Loss Account only for the year ended 31st March 2019 in the books of Sohan: Particulars Opening Stock Purchases Rs. 32000 171000 4000...

Study smarter with the SolutionInn App


