A sodium acetate solution contains 110 g of NaC 2 H 3 O 2 per 100 g
Question:
A sodium acetate solution contains 110 g of NaC2H3O2 per 100 g of water. Refer to Figure 13.6 and determine whether the solution is unsaturated, saturated, or supersaturated at each of the following temperatures.
(a) 50 °C
(b) 70 °C
(c) 90 °C.
Figure 13.6
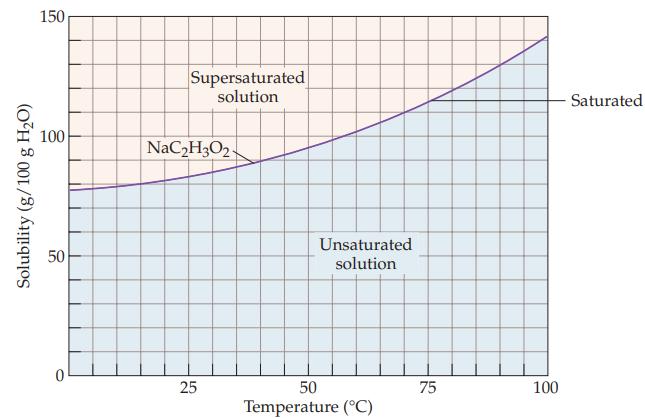
Transcribed Image Text:
Solubility (g/100 g H₂O) 150 100 50 0 Supersaturated solution NaC₂H302 25 Unsaturated solution 50 Temperature (°C) 75 100 Saturated
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a At 50 C the solubility of NaC 2 H 3 O 2 is about 97 g100 g water Since the solution ...View the full answer

Answered By

Lav Singh
I am a mathematics researcher working in the area of pure mathematics. I have published a few original articles in reputed journals. Besides research, I am passionate about teaching as well. I have been working on online-tutoring platforms for the past 4 years. I am also good at Latex and MS-word.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
On December 15, 20X2, you began as the new CFO of Trilogy Inc. Trilogy produces and manufactures stainless steel water bottles. You inherited the business (along with all of its problems) from your...
-
A sodium acetate solution contains 80 g of NaC 2 H 3 O 2 per 100 g water. Refer to Figure 13.6 and determine whether the solution is unsaturated, saturated, or supersaturated at each of the following...
-
A solution of 20.0 g KClO 4 in 500.0 g of water is brought to a temperature of 40 C. (a) Refer to Figure 14-10 and determine whether the solution is unsaturated or supersaturated at 40 C. (b)...
-
A room is 6 m by 5 m by 3 m. (a) If the air pressure in the room is 1 atm and the temperature is 300 K, find the number of moles of air in the room. (b) If the temperature rises by 5 K and the...
-
In what way do the adjustment and elimination entries for consolidation workpapers differ for the financial statement and trial balance approaches?
-
Barret Company has fixed costs of $10 million and faces a tax rate of 25%. The company accountant has advised you that the unit sales required to earn an after-tax profit of $600,000 is 100,000. If...
-
Explain how depreciation generates actual cash flows for a firm.
-
Stoney Saure, Votives, Inc.s accountant, must group the costs ofmanufacturing tealights. Indicate whether each of the following items should be classified as direct materials (DM), direct labor (DL),...
-
11.A simple LR circuit is connected to a battery at t = 0. The time instant at which rate of energy storage in inductor is half of power delivered by battery 2L (1) In 2 (3) In 2 (2) In (4) (4) In 3
-
How is it possible to exceed the saturation of a solution and produce a supersaturated solution?
-
The solubility of nitrous oxide is 0.12 g/100 g water at 20 C and 1.00 atm. What is the partial pressure required to dissolve 0.55 g of the gas in 100 g of water at 20 C?
-
Simon has recently discovered that his local town has a history which includes tales of murder, mystery and sightings of ghosts. He plans to start a small business running ghost tour evenings. Guests...
-
Current Attempt in Progress * Only those costs that differ across alternatives, i.e., relevant costs are to be considered. In incremental analysis, the only costs to be considered are variable costs....
-
If you are to write a code of ethics for a drug company ,will you include the provision of not using humans as guine pig for drug testing explaine your argument.
-
One of the operating system's objectives is the ability to evolve. Using two different operating system types as an example, explain TWO (2) reasons from each type of operating system on how the...
-
The p1,000 face value of Ayala Company bond has a coupon of 10% (paid semi-annually), matures in 4 years, and has a current price of p1,140. What is the bond's yield to maturity.
-
What are the three types of information a business owner might get from a projected profit and loss statement?
-
What tests must be met for an accrual basis taxpayer to deduct an expense?
-
Reduction in sales All of the above 29. Belt of an electric motor is broken, it needs a. Corrective maintenance b. Scheduled maintenance c. Preventive maintenance d. Timely maintenance. 30. The...
-
Calculating Returns and Variability Using the following returns, calculate the arithmetic average returns, the variances, and the standard deviations for X and Y. Returns Year 6% 18% 24 39 13 -6 -20...
-
Calculating Returns and Variability Youve observed the following returns on Crash-n-Burn Computer s stock over the past five years: 2 percent, 8 percent, 24 percent, 19 percent, and 12 percent. a....
-
Calculating Real Returns and Risk Premiums for Problem 9, suppose the average inflation rate over this period was 3.5 percent and the average T-bill rate over the period was 4.2 percent. a. What was...
-
Consider the following four brief cases of companies that are monitoring the business environment and deciding how to respond. 1. The company that introduced personal one-cup drip coffee makers to...
-
organizational leadership is considering buying a competitor and has asked the software security team to develop a plan to ensure the competitors point of sale system complies with organization...
-
How do database tables support advanced indexing techniques, such as clustered and non-clustered indexes, covering indexes, and index-organized tables, to accelerate query processing and facilitate...

Study smarter with the SolutionInn App


