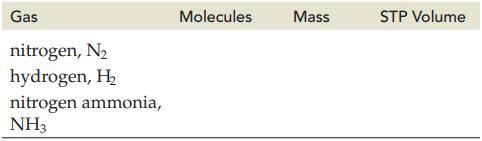
Given 1 mol of each gas listed, complete the following table. Gas nitrogen, N hydrogen, H nitrogen
Question:
Given 1 mol of each gas listed, complete the following table.
Transcribed Image Text:
Gas nitrogen, N₂ hydrogen, H₂ nitrogen ammonia, NH3 Molecules Mass STP Volume
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Gases Molecules Mass STP volu...View the full answer

Answered By

Anthony Ngatia
I have three academic degrees i.e bachelors degree in Education(English & Literature),bachelors degree in business administration(entrepreneurship option),and masters degree in business administration(strategic management) in addition to a diploma in business management.I have spent much of my life in the academia where I have taught at high school,middle level colleges level and at university level.I have been an active academic essays writer since 2011 where I have worked with some of the most reputable essay companies based in Europe and in the US.I have over the years perfected my academic writing skills as a result of tackling numerous different assignments.I do not plagiarize and I maintain competitive quality in all the assignments that I handle.I am driven by strong work ethics and a firm conviction that I should "Do Unto others as I would Like them to do to me".
4.80+
76+ Reviews
152+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Given 1 mol of each gas listed, complete the following table. Gas nitrogen, N oxygen, O2 nitrogen dioxide, NO Molecules Mass STP Volume
-
Complete the following table for an ideal gas: 0.500 mol ?K 2.00 atm 1.00L 0.300 atm 0.250 L 650 to ?L ? atm 27C 0.333 mo 350 K 585 mL 0.250 mo 295 K mol
-
Han Company has three products in its ending inventory. Specific per unit data at the end of the year for each of the products are as follows: Cost Replacement cost Selling price Selling costs Normal...
-
Allison is paid $1,520 per week. What is the amount of federal income tax withheld from Allisons paycheck under the following conditions? Use the percentage method table in the Appendix to this...
-
Data for The Freema Company are presented in E12-8. Instructions Prepare the entries to record: (a) The sale of non-cash assets. (b) The allocation of the gain or loss on realization to the partners....
-
Beach balls 1 ft in diameter have a thickness of 0.03 in. How much material would be needed to make 5000 beach balls?
-
A long straight wire carries a current of magnitude \(I\), and this current creates a magnetic field \(\vec{B}\). Derive an expression for the magnitude of the magnetic field a radial distance \(r\)...
-
Hindi Company has the following production data for April: units transferred out 40,000, and ending work in process 5,000 units that are 100% complete for materials and 40% complete for conversion...
-
1.Which of the following is an advantage of implementing just-in-time inventory management? Pick 1 option The frequency of raw materials deliveries is reduced Monthly finance costs incurred in...
-
Calculate the number of molecules in each of the following gases at STP. (a) 0.150 g of carbon monoxide, CO (b) 100.0 mL of hydrogen, H 2 .
-
Calculate the molar mass for each of the following gases given the STP density. (a) Ozone, 2.14 g/L (b) Silane, 1.43 g/L (c) Nitric oxide, 1.34 g/L (d) Freon-22, 3.86 g/L.
-
a. Write a possible sequence for an mRNA segment coding for apamine. b. Do you think apamine is synthesized in the form shown in Problem 11, or is it more likely a product of proteolytic cleavage of...
-
Can a cost be both a direct cost and an indirect cost?
-
M11 L1 - Activity 1: Static Electricity Investigation Lab Static Electricity Investigation Part 1: Electric Charges Lab Directions: Complete the Static Electricity Investigation Part 1: Electric...
-
Find the inverses of the matrices in Exercises 1-4. 8 3 [ 5 2 1. 2. 3 -3 8 3. [-9 [ -7 5. Verify that the inverse you found in Exercise 1 is correct. 6. Verify that the inverse you found in Exercise...
-
A company has two manufacturing departments Shaping and Painting. The company uses job - order costing and provides the following estimated overhead costs for the month of May: Beginning - of -...
-
1232 Mev. a) b) Find the ratio of cross sections for the following reactions, when the total CM energy is +pK) + (0) +pK +
-
Duo Co. has the following processing standards for its clerical employees: Number of hours per 1,000 papers processed ........ 150 Normal number of papers processed per year........ 1,500,000 Wage...
-
What kind of rays are X-rays?
-
Impairment of Debt Securities Cairo Corporation has municipal bonds classified as available for-sale at December 31, 2010. These bonds have a par value of $800,000, an amortized cost of $800,000, and...
-
Fair Value Measurement Presented below is information related to the purchases of common stock by Lilly Company during 2010. (a) What entry would Lilly make at December 31, 2010, to record the...
-
Fair Value Measurement Issues assume the same information as in E17-19 for Lilly Company. In addition, assume that the investment in the Woods Inc. stock was sold during 2011 for $195,000. At...
-
Let 1 f(z) = (2-1)(x-2) Find the Laurent expansion of f about z = 0 in the region {ze C: 1
-
Find all the complex numbers z such that z += 0. Is the set open or closed? {zC: z*+i=0}
-
For each of the following matrices A Maxn (R), test A for diagonal- izability, and if A is diagonalizable, find an invertible matrix Q and a diagonal matrix D such that Q-1AQ = D. (a) (63) 2 1 3 (b)...

Study smarter with the SolutionInn App


