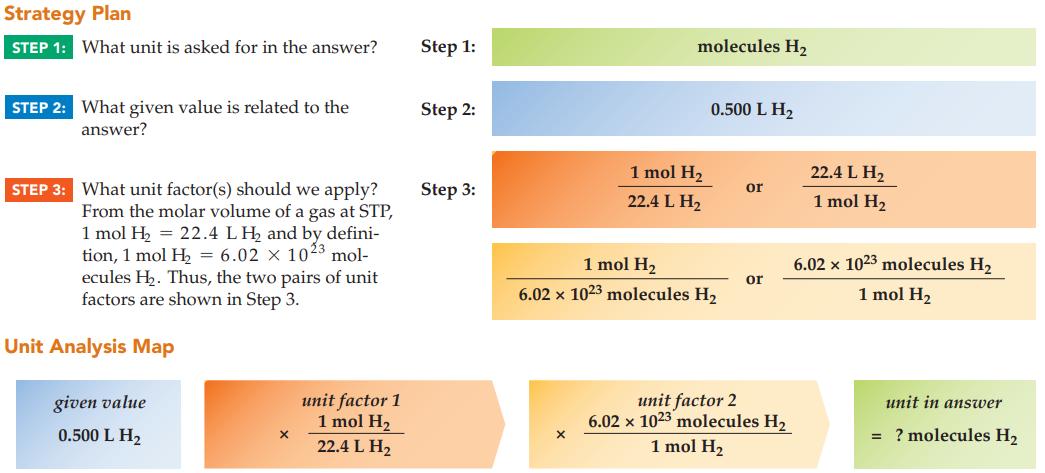
How many molecules of hydrogen gas, H 2 , occupy 0.500 L at STP? Strategy Plan STEP
Question:
How many molecules of hydrogen gas, H2, occupy 0.500 L at STP?
Transcribed Image Text:
Strategy Plan STEP 1: What unit is asked for in the answer? STEP 2: What given value is related to the answer? STEP 3: What unit factor(s) should we apply? From the molar volume of a gas at STP, 1 mol H₂ = 22.4 LH₂ and by defini- tion, 1 mol H₂ = 6.02 x 1023 mol- ecules H₂. Thus, the two pairs of unit factors are shown in Step 3. Unit Analysis Map given value 0.500 L H₂ X unit factor 1 1 mol H₂ 22.4 L H₂ Step 1: Step 2: Step 3: molecules H₂ X 1 mol H₂ 22.4 L H₂ 0.500 L H₂ 1 mol H₂ 6.02 x 1023 molecules H₂ or or unit factor 2 6.02 x 1023 molecules H₂ 1 mol H₂ 22.4 L H₂ 1 mol H₂ 6.02 x 1023 molecules H₂ 1 mol H₂ unit in answer = ? molecules H₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
We apply the unit factor 1 mol H224 LH t...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Strong and Tall's Inc weight gain program headline says, "Gain up to 20 kg in 60 days or we will give you your money back." The 2 types of headline this ad shows is: a. command & benefit b. command &...
-
Nitrogen and hydrogen combine at high temperature, in the presence of a catalyst, to produce ammonia.? N2(g)+3H2(g)---->2NH3(g) Assume 4 molecules of nitrogen and 9 molecules of hydrogen are...
-
How many molecules of H2 are needed to react with 29 molecules of N2 to make ammonia if the balanced chemical equation is N2 + 3H2 2NH3?
-
Write the C++ code for a function that receives an integer, a double number, and the address of a double variable from the calling statement. The function should multiply the integer by the double...
-
Brasswood Corporation reports the following amounts in its 2012 financial statements: Instructions(a) Compute the December 31, 2012, balance in stockholders?? equity.(b) Compute the debt to total...
-
Because of the time that many people spend indoors, there is a concern about the health risk of being exposed to harmful fungi that thrive in buildings. The risk appears to increase in damp...
-
A particle of mass \(9.1 \times 10^{-31} \mathrm{~kg}\) and carrying an unknown quantity of charge is shot at a velocity of \(2.0 \times 10^{4} \mathrm{~m} / \mathrm{s}\) to the right and enters the...
-
Evans Industries wishes to select the best of three possible machines, each of which is expected to satisfy the firms ongoing need for additional aluminum-extrusion capacity. The three machinesA, B,...
-
123456789 10 What is the need of MIS? What are the objectives of MRP? JIT introduced in which country? Advantages of TQM. Discuss about six sigma. Define various levels of CMM. What is mean by supply...
-
Given Avogadros number of shotput balls, which of the following is the best estimate of the total mass: an automobile, the Empire State building, or Earth?
-
How many molecules of ozone are in one mole of gas?
-
Locate the discontinuities of the function and illustrate by graphing. 30. y = In(tan?x) 29. y - I+elis
-
Practice Set #4 Having recorded, posted (to the General Ledger) and completed the '1-31-23 Trial Balance' worksheet with the unadjusted balances (Practice Set #3), we are now going to prepare the...
-
Discuss, how AI Tech (artificial intelligence technology) could help financial advisers in the client interview process? (200 words)
-
2. The Council on Postsecondary Education will limit tuition increases at Kentucky Colleges to 3 percent. This government intervention is an example of (Quota/ Price Ceiling/ Price Floor). Show it on...
-
When financial leverage is used, what is the potential impact on before-tax cash flow and the current before-tax yield to the equity position? 3. Explain the meaning of the loan-to-value ratio from a...
-
LKO is a somewhat mature company but is seeking growth capital. Right now LKO Co. has EBIT of 10, CAPEX requirements of 5, Depreciation of 3 and negligible changes in working capital year-to-fear....
-
Explain the possible causes for direct materials price and direct materials usage variances. Who in the organization normally has influence over or responsibility for these variances?
-
Which of the ocean zones shown would be home to each of the following organisms: lobster, coral, mussel, porpoise, and dragonfish? For those organisms you identify as living in the pelagic...
-
Gross Profit Method Castlevania Company lost most of its inventory in a fire in December just before the year-end physical inventory was taken. The corporations books disclosed the following....
-
Gross Profit Method You are called by Kevin Garnett of Celtic Co. on July 16 and asked to prepare a claim for insurance as a result of a theft that took place the night before. You suggest that an...
-
Gross Profit Method Sliver Lumber Company handles three principal lines of merchandise with these varying rates of gross profit on cost. Lumber.......................25%...
-
Lindal Corporation, organized in 2023, immediately filed an election for S corporation status under the rules of Subchapter S. What is the maximum amount of passive investment income that Lindal will...
-
if assets are 1 1 5 , 0 0 0 , owner investments are 2 9 , 0 0 0 , loss of 2 5 , 5 0 0 and owner withdrawals are 6 , 9 0 0 . what are the liabilities
-
What is immediate, up-to-date information? What is Real-time systems Information governance?

Study smarter with the SolutionInn App


