Question: Identify five functional groups in thyroxine (thyroid hormone). HO- I I I I -CH-CH-C-OH NH
Identify five functional groups in thyroxine (thyroid hormone).
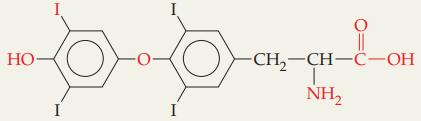
HO- I I I I -CH-CH-C-OH NH
Step by Step Solution
3.54 Rating (144 Votes )
There are 3 Steps involved in it
Thyroxine contains the ... View full answer

Get step-by-step solutions from verified subject matter experts


