Identify nuclide X in the following nuclear reactions that represents the radioactive decay product from (a) Beta
Question:
Identify nuclide X in the following nuclear reactions that represents the radioactive decay product from
(a) Beta emission and
(b) Positron emission.
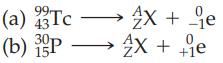
Transcribed Image Text:
99 43 (a) Tc (b) P 30 15 0 X + e → 4X + 0 +16
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a 99...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Identify X in the following nuclear reaction (a) 1H + 9Be X + n; (b) 12C + 1H X; (c) 15N + 1H 4He + X.
-
Terence purchased a whole life policy in 1977, which he later transferred to his wife Gertrude in 1989. What group would Gertrude's policy fall under for tax purposes? a) G1 b) G2 c) G3 d) G4
-
Use drawings to complete the following nuclear reactions (orange circles represent neutrons and blue circles represent protons). Once you have completed the drawings, write the nuclide symbols under...
-
Consider a spherical fluid particle in an inviscid fluid (no shear stresses). If pressure and gravitational forces are the only forces acting on the particle, can they cause the particle to rotate?...
-
Basic information relating to a new machine purchased by Deloise Company is presented in E9-5. Instructions Using the facts presented in E9-5, compute depreciation using the following methods in the...
-
Find the reference angle and the exact function value if they exist. cos ( -180)
-
A long wire that is at rest in the Farth reference frame initially carries no current. Observer \(\mathrm{E}\) in this reference frame measures linear charge densities of \(\lambda_{\mathrm{En}}=\)...
-
Munoz Sporting Equipment manufactures baseball bats and tennis rackets. Department B produces the baseball bats, and Department T produces the tennis rackets. Munoz currently uses plant wide...
-
In the database you have built, you need to create a relationship between two fields in two tables. Field 1 in Table A has the Number Data Type and Field 1 in Table B has the Currency Data Type. Will...
-
Which type of natural radiation corresponds to each of the following descriptions? (a) Stopped by human skin and requires heavy cloth shielding (b) Penetrates human skin and requires aluminum...
-
Write a balanced nuclear equation for each of the following radioactive decay reactions. (a) Radon-222 decays by alpha and gamma emission. (b) Barium-133 decays by electron capture.
-
Compare Walmart and Amazon's business models and business strategies.
-
Before we can _____________ your property, we must _____________ it. a. ensure, appraise b. assure, apprise c. insure, appraise For the above sentence, write the correct letter in the space provided.
-
Of the _____________ viewer comments about our commercial we received, _____________ were negative. a. 68, nine b. 68, 9 c. sixty-eight, nine For the above sentence, write the correct letter in the...
-
As an intern at VisibleThread, the compliance and clarity experts, you and several other interns have been assigned the task of researching the history of the plain writing/plain language movement....
-
The State _____________ in Denver is the _____________ government building in Colorado. a. Capital, premiere b. Capitol, premier c. Capitol, premiere For the above sentence, write the correct letter...
-
The following e-mail from Avianca Keller, the vice president of employee relations, seeks to help supervisors and managers write safe and helpful performance reviews. Your Task. Analyze the...
-
Gleason and Company is planning to invest $5 million in a new, stand-alone project. Before depreciation, it expects this project to yield a positive cash flow of $1.4 million each year for five...
-
As economic conditions change, how do banks adjust their asset portfolio?
-
Based on the data in Exercise 10-12, determine the depreciation for the Kubota tractor for each of the first two years, using the sum-of-the-years-digits depreciation method. Round to the nearest...
-
Based on the data in Exercise 10-13, determine the depreciation for the storage tank for each of the first two years, using the sum-of-the-years-digits depreciation method. Round to the nearest...
-
Based on the data in Exercise 10-14, determine the depreciation for the sandblasting equipment for each of the first two years, using the sum-of-the-years-digits depreciation method. Round to the...
-
Problem 1 [40 points]: A thermal gravimetric analyzer (TGA) is a common instrument used in materials science laboratories. In this instrument, a sample is placed on a balance that is suspended inside...
-
With the following information calculate the cash conversion cycle the gross profit margin of the company is 30% Sales $ 350,000.00 Cash Balance $ 55,000.00 Accounts Receivables $ 65,000.00 Accounts...
-
Coates writes about the precarious position of the Black body in America. What are some specific examples of the dangers that face the Black body?

Study smarter with the SolutionInn App


