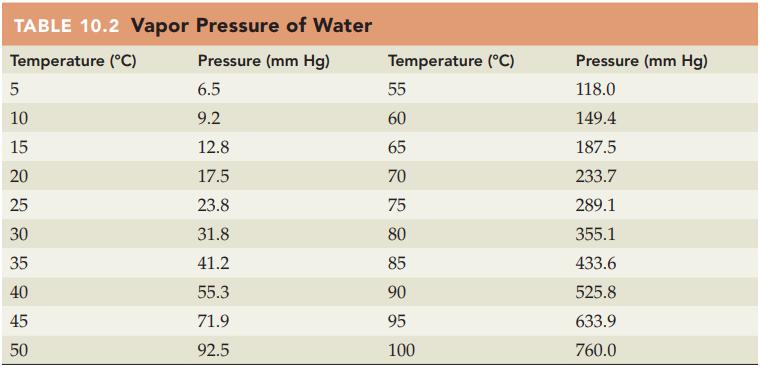
Refer to Table 10.2 and state the vapor pressure for water in mm Hg at each of
Question:
Refer to Table 10.2 and state the vapor pressure for water in mm Hg at each of the following temperatures:
(a) 25 °C
(b) 50 °C.
Table 10.2
Transcribed Image Text:
TABLE 10.2 Vapor Pressure of Water Temperature (°C) Pressure (mm Hg) 6.5 9.2 5 10 15 20 25 30 35 40 45 50 12.8 17.5 23.8 31.8 41.2 55.3 71.9 92.5 Temperature (°C) 55 60 65 70 75 80 85 90 95 100 Pressure (mm Hg) 118.0 149.4 187.5 233.7 289.1 355.1 433.6 525.8 633.9 760.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Temperature C a 25 Co Vapor ...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to Table 10.2 and state the vapor pressure for water in atm at each of the following temperatures: (a) 75 C (b) 100 C. Table 10.2 TABLE 10.2 Vapor Pressure of Water Temperature (C) Pressure (mm...
-
You are supplied with a 4.12 % concentration (by weight) hydrogen peroxide solution. Assume the density of your solution is 1.00 g/mL. a. Write a balanced equation for the reaction of aqueous...
-
The HaberBosch process for the production of ammonia is one of the key industrial processes in developed countries. N 2 (g) + 3 H 2 (g) 2 NH 3 (g) (a) Calculate r G for the reaction at 298 K, 800...
-
The following data relating to direct materials cost for March of the current year are taken from the records of Play Tyme Inc., a manufacturer of plastic toys: Quantity of direct materials used...
-
Is getting a certificate to buy something you would have bought anyway equivalent to getting money? What if it was to buy something you would not have bought anyway?
-
If a large force is applied to an object, does it necessarily follow that work in done on it? \(\bullet\)
-
Laser Cast, Inc., manufactures color laser printers. Model A200 presently sells for $400 and has a total product cost of $320, as follows: Direct materials $230 Direct labor ...... 60 Factory...
-
1. Letter of the word INDIANOIL are arranged in all possible ways. The number of permutations in which A, I, O occur only at odd places, is : (a) 720 2. (b) 360 (c) 240 (d) 120 How many different...
-
Which of the following liquids has the higher vapor pressure at 50 C: alcohol or mercury
-
Which of the following liquids has the higher vapor pressure at 25 C: water or mercury?
-
Plot the variation in the bus 2 phase \ (a, b, c\) voltage magnitudes during a single line-to-ground fault at bus 2 as the fault reactance is varied from 0 to 0.30 per unit in 0.05 per-unit steps the...
-
A box was found to have a volume of 9.56 cm3. What is the boxes volume in units of cubic inches. use dimensional analysis. Show all work please
-
Discuss the reasons why the net present value investment appraisal method is preferred to other investment appraisal methods such as payback, return on capital employed and internal rate of return.
-
1. What is Opportunity Cost of Credit 2. What are the other charges and their Effect on Cost of Credit
-
Most borrowers only pay attention to the monthly payment when taking out a loan. Why do you think this is? How can this benefit the lender?
-
Introduce the case study including challenges (global trade is more difficult for small business than large) in the environment (back, country, finance sector) explained and the problem being solved...
-
Generally, the value chains involved in providing a given product or service can be from multiple industries that can be interconnected in complex ways. One example is the auto industry where the...
-
The following information is available for Partin Company: Sales $598,000 Sales Returns and Allowances 20,000 Cost of Goods Sold 398,000 Selling Expense 69,000 Administrative Expense 25,000 Interest...
-
List the six steps in estimating a cost function on the basis of an analysis of a past cost relationship which step is typically the most difficult for the cost analyst?
-
When using the high-low method, should you base the high and low observations on the dependent variable or on the cost driver?
-
Describe three criteria for evaluating cost functions and choosing cost drivers.
-
List and describe all the ethical theories. Give an example (for each) of a situation where you think application of the theory would or would not be justified. 15 pts What are your takeaways from...
-
Reva, a stellar student, is upset to learn that Joan, the class entrepreneur, secretly copied her detailed Civil Litigation notes. After months of negotiating with Joan prove fruitless, Reva sues...
-
Although there are differences between Canada and the United States, the ethical principles are the same. Refer to the "Code of Ethics for Medical Assistants" (fig.6-1) on page 79 Kinn's textbook. Do...

Study smarter with the SolutionInn App


