Refer to the periodic table and determine the atomic number and atomic mass for sulfur. 2 3
Question:
Refer to the periodic table and determine the atomic number and atomic mass for sulfur.
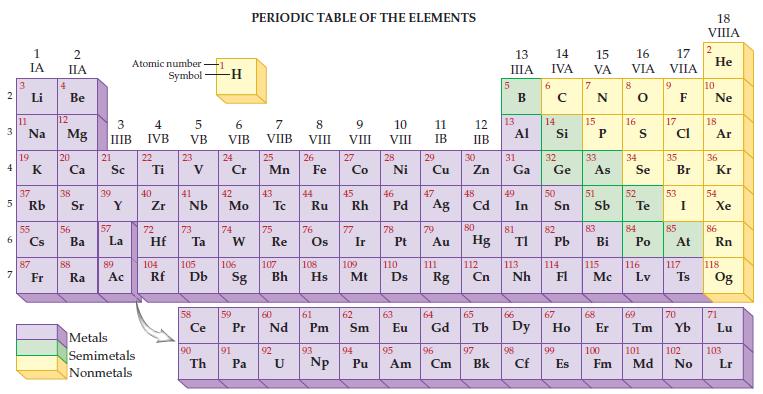
Transcribed Image Text:
2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21 Sc 39 57 Y La 89 Ac Atomic number Symbol Metals Semimetals Nonmetals 4 IVB 22 Ti 40 Zr 5 VB 72 23 41 Nb 73 Hf Ta Ce 90 -1 Th -H 6 VIB 24 Cr 42 Mo 74 W 59 91 PERIODIC TABLE OF THE ELEMENTS 7 VIIB Pa 25 Mn 43 75 Re 60 P Pr Nd 92 8 VIII U 26 44 Fe Ru 76 61 Pm 93 9 VIII 104 105 106 107 108 109 110 Rf Db Sg Bh Hs Mt D Np 27 Co 45 Rh 77 Ir 62 Sm 94 10 VIII Pu 28 Ni 46 78 47 Pd Ag 63 11 IB 29 95 Cu 79 Au 111 64 Eu Gd Rg 96 Am Cm 12 IIB 30 Zn 48 Cd 80 Hg 112 Cn 65 97 Bk 13 IIIA 13 31 Ga Al 49 81 In 66 TI 113 Nh 98 Cf 6 14 15 16 IVA VA VIA 14 Si 32 Ge 50 82 Pb 114 67 99 E Es 15 33 As 51 83 Bi 115 Mc 68 8 16 34 Se 52 S Te 84 Po 116 Lv 69 17 VIIA 9 17 Cl 35 53 85 Br I At 117 70 Ts 100 101 102 Fm Md No 18 VIIIA 2 He 10 Ne 18 Ar 36 Kr 54 Xe 86 Rn 118 Og 71 Lu 103 Lr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
16 ...View the full answer

Answered By

Sandip Agarwal
I have an experience of over 4 years in tutoring. I have solved more than 2100 assignments and I am comfortable with all levels of writing and referencing.
4.70+
19+ Reviews
29+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to the periodic table and determine the atomic number and mass number listed for element 61, promethium. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB...
-
What would you suggest to be done and which principles are important to you in this decision? How would you think about the greatest good in this case? As a leader what are your duties and who are...
-
The electronics industry manufactures transistors using arsenic diffusion. Refer to the periodic table and predict an element that may substitute for arsenic. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55...
-
Each table of values gives several points that lie on a line.(a) What is the x-intercept of the line? The y-intercept?(b) Which equation in choices AD corresponds to the given table of values?(c)...
-
What are the two dimensions of a total quality management (TQM) program? Why is TQM being used in business practice?
-
What did Brown and BWNS do well in this situation? What could have been done better? MINI CASE Reggie Brown, B&W Nuclear Service Companys (BWNS) project manager for Nita Light and Powers Green Meadow...
-
Mountain climbers know that it is harder to hike down a hill than to walk on level ground. Why is this so?
-
Suppose that the following four fundsall with committed capital of $100Mhave combined to form a syndicate to invest in Newco: (I) ABC Fund, management fees of 2.5 percent per year of committed...
-
Camila is working part time earning $925 gross income per month. If Camila has 30.6% in tax deductions, what is her net income?
-
Sodium has only one stable isotope. What is the mass of the Na-23 stable isotope?
-
Refer to the periodic table on the inside cover of this text and determine the atomic number and atomic mass for iron. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra...
-
So you want to ban smoking in all eating establishments? Well, you go right ahead and do that little thing. And when the 40 percent of smokers stop eating out, the restaurants can do one of two...
-
What is meant by the term benchmarking, and why is it important in human resources management?
-
In what sense are all healthcare executives human resources managers? How can executives best prepare to perform well in this HR function?
-
Given two equally qualified job applicantsone from inside and one from outside the organizationhow would you go about deciding which one to hire?
-
What is the policy of the organization on compensating employees at market rates? Is there an explicit policy to pay below market, at market, or above market? Does the approach vary by the type of...
-
What are the benefits and drawbacks of using nonstandard employees, standard employees, and outsourced staff?
-
Explain the relationship between the Supremacy Clause and the doctrine of federal preemption.
-
Interest Compounded Annually. When P dollars is invested at interest rate i, compounded annually, for t years, the investment grows to A dollars, where A = P(1 + i) t . Trevor's parents deposit $7800...
-
Assume that it is now July of year 1, and the brothers are developing pro forma financial statements for the following year. Further, assume that sales and collections in the first half-year matched...
-
Assume now that it is several years later. The brothers are concerned about the firm's current credit terms, which are now net 30, which means that contractors buying building products from the firm...
-
Under the current credit policy, what is the firm's days sales outstanding (DSO)? What would the expected DSO be if the credit policy change were made?
-
A liquIn the U.S., excess reserves held at the central bank pay interest to the DIid asset can be converted to cash quickly, but will require a discount from market value
-
The identification portion for Marriott hotels of the project is valued at 5 percent of your final course grade, and consists of gathering financial information (financial reports, financial ratios)...
-
When Sprint and Nextel merged in 2005, the combined stock lost 76 percent of its value by 2021 at which point SoftBank agreed to purchase the company. SoftBank stock dropped 20 percent the week after...

Study smarter with the SolutionInn App


