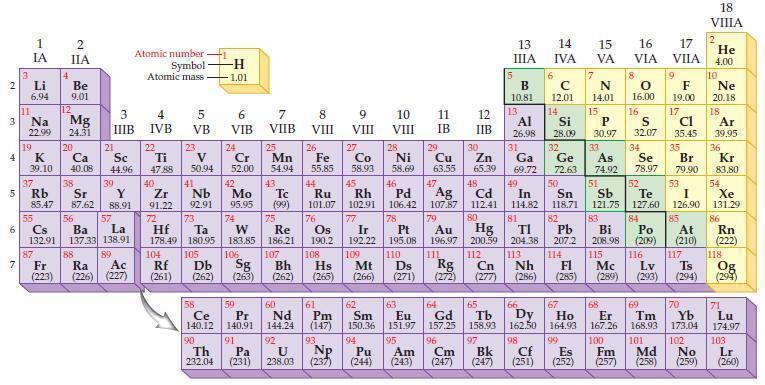
Refer to the periodic table and predict which of the following ions are isoelectronic with the noble
Question:
Refer to the periodic table and predict which of the following ions are isoelectronic with the noble gas xenon:
(a) Cs+
(b) Cl–
(c) La3+
(d) Se2–.
Periodic Table:
Transcribed Image Text:
2 3 4 15 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 Rb R 4 87 2 IIA Be 9.01 12 Mg 24.31 K Ca Sc 39.10 40.08 44.96 20 38 21 3 IIIB Sr Y 85.47 87.62 88.91 88 39 55 56 La Cs Ba 132.91 137.33 138.91 57 89 Fr Ra Ac (223) (226) (227) Atomic number Symbol Atomic mass- 4 IVB 22 Ti 47.88 40 Zr 2 91.22 72 5 VB 104 23 V 50.94 41 73 Hf Ta 178.49 180.95 105 Rf Db (261) (262) -H -1.01 6 VIB Nb Mo 92.91 95.95 90 24 Cr 52.00 42 74 106 Sg (263) 58 Pr Ce 140.12 140.91 91 59 7 VIIB Th Pa 232.04 (231) 25 W Re 183.85 186.21 Mn 54.94 43. Tc (99) 75 107 Bh (262) 60 Nd 144.24 92 U 238.03 8 VIII 26 Fe 55.85 76 Os 190.2 61 Pm (147) 93 Np 9 VIII (237) 10 VIII 11 IB 12 IIB 27 28 29 Zn Co Ni Cu 58.93 58.69 63.55 65.39 44 49 45 46 48 Ru Rh Pd Cd In 101.07 102.91 106.42 107.87 112.41 114.82 Ag 77 78 79 80 Ir Pt 192.22 195.08 109 110 Hs Mt Ds Rg Cn (265) (266) (271) (272) (277) Au Hg 196.97 200.59 111 112 108 47 30 5 13 ΠΙΑ B 10.81 13 6 14 IVA с 12.01 14 81 82 TI Pb 204.38 207.2 113 114 Nh Fl (286) (285) 7 66 Ho 62 63 64 65 Sm Eu Gd Tb Dy 150.36 151.97 157.25 158.93 162.50 164.93 99 94 95 96 97 Pu Am Cm Bk (244) (243) (247) (247) 15 VA 67 N 14.01 Ne 20.18 18 Al Si S Ar 32.07 34 39,95 36 31 Kr 83.80 54 53 26.98 28.09 32 33 Ga Ge As Se 69.72 72.63 74.92 78.97 50 51 52 Sn Sb Te I Xe 118.71 121.75 127.60 126.90 131.29 83 84 85 86 Bi Po At Rn 208.98 (209) (210) (222) 115 116 117 118 Lv Ts (289) (293) (294) Mc Og (294) 15 16 17 VIA VILA P 30.97 8 O 16.00 9 F 19.00 17 Cl 35.45 16 35 68 69 70 Er Tm Yb 167.26 168.93 173.04 100 101 102 Cf Es Fm Md No (251) (252) (257) 98 (258) (259) Br 79.90 18 VIIIA 2 He 4.00 10 71 Lu 174.97 103 Lr (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a Cs is isoelectronic with ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to the periodic table and predict which of the following ions are isoelectronic with the noble gas argon. (a) Al 3 + (b) Ca 2 + (c) S 2 (d) N 3 . Periodic Table: 2 3 4 10 6 3 7 11 Li 6.94 1 IA...
-
Refer to the periodic table and predict which of the following ions are isoelectronic with the noble gas argon: (a) K + (b) Br (c) Ca 2+ (d) O 2 . 2 3 4 5 6 7 Li 6.94 11 1 IA Na 22.99 19 37 55 4...
-
Refer to the periodic table and predict which of the following ions are isoelectronic with the noble gas krypton. (a) K + (b) Sr 2+ (c) Cl (d) Se 2 .
-
How are the square numbers embedded in Pascals triangle?
-
U.S. Government-wide Annual Report. Obtain the most recent audited annual financial report of the U.S. government. It is available from the Government Accountability Office (GAO). Required a. Did the...
-
White Farm Supplys records for the first three months of its existence show purchases of commodity Y2 as follows: The inventory of commodity Y2 at the end of October using FIFO is valued at $36,390....
-
Which of the following statements is false? (a) A negative cash flow can occur in a year in which net income is positive. (b) An increase in accounts receivable represents accounts not yet collected...
-
The production supervisor of the Machining Department for Nell Company agreed to the following monthly static budget for the upcoming year: Nell Company Machining Department Monthly Production Budget...
-
Find the explicit GS of: xy=y+x + y.
-
What is the term for the elements in the series that follows element 57?
-
Predict the common ionic charge for Group VIIA/17 elements; Group VIA/16 elements; Group VA/15 elements.
-
What is an ANOVA?
-
Suppose you draw three balls without replacement from a bag of balls numbered 110. How many different possible combinations sample balls you have?
-
Suppose the lifetime of a television picture tube is distributed exponentially with a standard deviation of 1,400 h. Find the probability that the tube will last: (a) More than 3,000 h (b) Less than...
-
A random sample of 75 observations from a population yielded the following summary statistics: Construct a 95 % confidence interval for the population mean m. x=1,270 x = 21,520
-
From time to time, managers may be faced with the need to change the internal rules of the game within their MNE. What skills and capabilities may be useful in achieving this?
-
The data entry operation in a large computer department claims that it gives its customers a turnaround time of 6.0 h or less. To test this claim, one of the customers took a sample of 36 jobs and...
-
Analyze the following data by using a multiple regression computer software package to predict y using x1 and x2. Notice that x2 is a dummy variable. Discuss the output from the regression analysis;...
-
What are the 5 Cs of marketing channel structure?
-
The residual value is the estimated fair value of the leased property at the end of the lease term. (a) Of what significance is (1) an unguaranteed and (2) a guaranteed residual value in the lessees...
-
How should changes in the estimated unguaranteed residual value be handled by the lessor?
-
Describe the effect of a bargain-purchase option on accounting for a capital-lease transaction by a lessee.
-
3. The 5200 chickens at Colonel Thompson's Ranch have a mean weight of 1850 g. with a standard deviation of 150g. Find the number of chickens weighing between 1750 and 1900 g.
-
Floretta Sutphin has worked for several years as an employee of a Canadian public company that has annual gross revenues of $190 million. In September 2021, Floretta was granted options to acquire...
-
Costco, the wholesale corporation, is the company of my research. While preparing to review the statement of cash flows, I thought it was going to be the usual, "profit, profit, profit" that I have...

Study smarter with the SolutionInn App


