Refer to the periodic table and write the predicted electron configuration for each of the following ions
Question:
Refer to the periodic table and write the predicted electron configuration for each of the following ions using core notation:
(a) Fe3+
(b) Se2–.
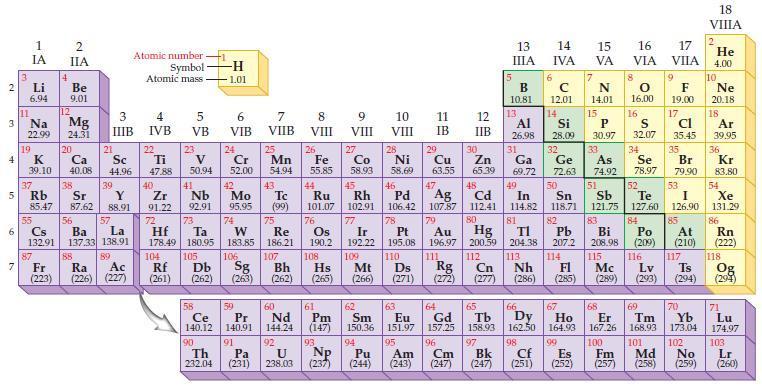
Transcribed Image Text:
2 3 4 5 6 7 Li 6.94 11 1 IA Na 22.99 19 37 55 4 2 IIA 87 Be 9.01 12 K Ca 39.10 40.08 Mg 24.31 20 38 21 Rb Sr Y 85.47 87.62 88.91 88 Sc 44.96 3 4 IIIB IVB 39 56 Cs La Ba 132.91 137.33 138.91 57 89 Atomic number Symbol Atomic mass Fr Ra Ac (223) (226) (227) 22 Ti 47.88 40 Zr 91.22 72 104 5 VB Rf (261) 23 V 50.94 41 Nb 92.91 Hf Ta 178.49 180.95 73 105 58 Ce 140.12 90 -H 1.01 Th 232.04 6 VIB 24 Cr 52.00 106 7 VIIB 42 Tc Mo 95.95 (99) 74 25 Db Sg Bh (262) (263) (262) 59 Pr 140.91 91 Mn 54.94 75 Re 183.85 186.21 Pa (231) 43 107 60 Nd 144.24 8 VIII 26 Fe 55.85 44 Ru 101.07 76 Os 190.2 9 VIII 92 U 238.03 (237) NP 93 45 Rh 102.91 77 10 VIII 27 28 Co Ni 58.93 58.69 63.55 65.39 Cu Zn Ir 192.22 109 62 61 Sm Pm (147) 150.36 94 Pu Am (244) (243) 46 Pd 106.42 78 63 Eu 151.97 95 11 IB 29 12 IIB 47 30 5 48 Cd 112.41 80 Hg 200.59 13 IIIA 32 33 34 Ge As Se 72.63 74.92 78.97 50 53 54 Ag 51 52 Sn Sb Te I Xe 118.71 121.75 127.60 126,90 131.29. 82 85 107.87 79 Pt Au 195.08 196.97 110 111 Ds 83 84 Pb Bi Po 207.2 208.98 (209) 108 204.38 113 Nh 115 116 112 Cn Rg 114 Fl Mc Hs Mt (265) (266) (271) (272) (277) (286) (285) (289) (293) (294) Lv B с 10.81 12.01 14 15 IVA VA N 14.01 15 13 14 Al Si P 26.98 28.09 30.97 31 6 Ga 69.72 49 In 114.82 81 7 64 66 65 67 Gd Tb Dy Ho 157.25 158.93 162.50 164.93 96 97 Cm Bk (247) (247) 16 VIA 8 0 16.00 16 S 32.07 17 VIIA 68 69 Er Tm 167.26 168.93 98 99 100 101 Cf Es Fm Md (251) (252) (257) (258) 9 F 19.00 17 Cl 35.45 35 Br 79.90 At (210) 18 VIIIA 2 117 He 4.00 10 Ne 20.18 18 Ar 39.95 36 Kr 83.80 86 Rn (222) 118 Ts Og (294) 70 71 Yb Lu 173.04 174.97 102 103 No Lr (259) (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
We refer to the periodic table to recall the blocks of elements so that we can write the electron co...View the full answer

Answered By

Lilian Nyambura
Hi, am Lilian Nyambura, With extensive experience in the writing industry, I am the best fit for your writing projects. I am currently pursuing a B.A. in Business Administration. With over 5 years of experience, I can comfortably say I am good in article writing, editing and proofreading, academic writing, resumes and cover letters. I have good command over English grammar, English Basic Skills, English Spelling, English Vocabulary, U.S. English Sentence Structure, U.K. or U.S. English Punctuation and other grammar related topics. Let me help you with all your essays, assignments, projects, dissertations, online exams and other related tasks. Quality is my goal.
4.80+
378+ Reviews
750+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to the periodic table and write the predicted electron configuration for each of the following ions using core notation: (a) Cd 2+ (b) P 3 . Periodic Table: 2 3 4 15 6 7 3 11 Li 6.94 1 IA Na...
-
Refer to the periodic table and write the predicted electron configuration for each of the following negative ions using core notation. (a) Br (b) Te 2 (c) As 3 (d) O 2 . Periodic Table: 2 3 4 10...
-
Refer to the periodic table and write the predicted electron configuration for each of the following negative ions using core notation. (a) F (b) S 2 (c) N 3 (d) I . Periodic Table: 2 3 4 10 6 3...
-
The stress field (15.1.7) for the screw dislocation produces no tangential or normal forces on a cylinder of finite radius with axis along the dislocation line (z-axis). However, show that if the...
-
Fund balance with U.S. Treasury. One amount is missing in the following trial balance of proprietary accounts, and another is missing from the trial balance of budgetary accounts of a certain agency...
-
On November 6 of Year 1, the company purchased inventory (on account) from a supplier located in Indonesia. The purchase price is 100,000,000 Indonesian rupiah. On November 6, the exchange rate was...
-
What is net cash flow from investing activities? (a) $14,000 (b) ($14,000) (c) $21,000 (d) ($16,000) The following information is available for Jacquis Jewelry and Gift Store: Net income Depreciation...
-
On January 1, 201X, Acorn Corporation issued $600,000 of 10%, 20-year bonds for $509,580, yielding a market rate of 12%. Interest is paid on July 1 and December 31. Acorn uses the interest method to...
-
8) You obtain a $1000 treasure note with a 6% annual rate, paid semi-annually, with a maturity in 5 years. How much interest will you earn?
-
Predict the next ion in the isoelectronic series: Se 2 , Br , Kr, Rb + , Sr 2+ .
-
What is the term for the elements in the series that follows element 57?
-
A hot steel ball is dropped into a cold aluminum cup containing some water. ( Assume the system is an isolated.) If the ball loses 400 J of heat, what can be said according to calorimetry?
-
A consumer rights organization tests a new car to estimate the cars average gasoline mileage. Because its budget is limited, the organization can test only 25 cars. The standard deviation of the cars...
-
An art dealer at an auction believes that the bid on a certain painting will be a uniformly distributed random variable between $500 and $2,000. (a) What is the probability density function for this...
-
A recent poll shows that 53 % of the voters interviewed strongly support the incumbent and are willing to vote for her in the coming election. The poll was taken by asking 1,000 voters. Estimate the...
-
Suppose the cost of sampling is 50 cents per observation. If the population has zero variance, large a sample should be collected to estimate mean of the population?
-
Suppose the random variable X is best approximated by an exponential distribution with = 8. Find the mean and the variance of X.
-
The Minitab output displayed here is the result of a multiple regression analysis with three independent variables. Variable x1 is a dummy variable. Discuss the computer output and the role x1 plays...
-
Cassandra Casey operates the Futuristic Antique Store. She maintains subsidiary ledgers for accounts payable and accounts receivable. She presents you with the following information for October 2019:...
-
Identify the lease classifications for lessors and the criteria that must be met for each classification.
-
Outline the accounting procedures involved in applying the direct-financing method.
-
Outline the accounting procedures involved in applying the operating method by a lessor.
-
You are discussing the investment of your pen- You are die with Sam in Lee, a representative of Red Rock Financial Services. Samson is talk- ing over the various investment options (similar to those...
-
Determine the type of immigration laws or visa requirements that need to be met to work in Germany.?
-
Does the charter of rights and freedoms allow a Canadian Citizen to defame another individual under the guarantee of Freedom of Expression?

Study smarter with the SolutionInn App


