Refer to the values in Figure 12.9 and calculate the electronegativity difference in a HP bond. Figure
Question:
Refer to the values in Figure 12.9 and calculate the electronegativity difference in a H—P bond.
Figure 12.9
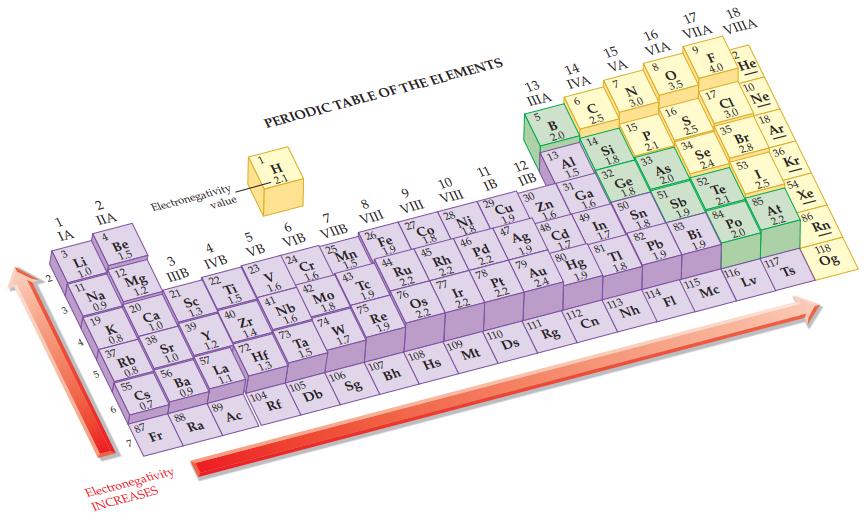
Transcribed Image Text:
1 IA S Li 1.0 11 2 IIA 4 Na 0,9 19 Be 1.5 12 0.8 37 Mg 1.2 6 20 Rb 0.8 55 Ca 1.0 38 3 Electronegativity 0.7 87 IIIB 21 Sr 1.0 56 Fr Sc Electronegativity INCREASES 39 Ba 0.9 88 1,3 value 4 IVB 22 Y 1.2 57 Ra Ti 1.5 40 La 1.1 89 5 VB 23 Zr 14 72 Ac PERIODIC TABLE OF THE ELEMENTS H 2.1 V 1.6 41 Hf 13 104 6 VIB 24 Nb 1.6 73 Rf Cr 1.6 42 Ta 15 7 VIIB 105 25 Mo 1.8 74 Db Mp 1.5 43 W 1.7 8 VIII 106 26 Tc 1.9 75 Sg Fe 19 44 Re 1.9 107 9 VIII 27 Ru 2.2 76 Bh 18 45 Os 2.2 10 VIII 108 28 Rh 22 77 Hs N 46 Ir 22 109 11 IB 29 Pd Mt 22 78 Cu 19 47 Pt 22 110 12 IIB 13 ΠΙΑ 5 130 Ag CE FOR 1.9 Ds 79 B 2.0 Zn 1.6 13 48 Au 111 24 14 IVA 6 AI 1.5 31 Cd 17 Rg 80 с 2.5 14 Ga 1.6 49 112 Hg 15 VA Cn Si 18 In 17 7 32 81 N 3.0 Ge 1.8 F 15 50 113 1.8 16 VIA 8 P 21 Nh 33 Sn 18 82 0 3.5 16 As 20 51 114 Pb 19 17 VIIA FI S 2.5 Sb 1.9 9 34 83 Se 24 Bi 19 F 12 4.0 He 17 52 115 18. VIIIA CL Mc 3.0 35 Te 2.1 84 10 Br 2.8 53 Po 20 116 Ne 18 I 25 85 Lv 36 Kr At 2.2 117 (54 Xe श Ts 86 Rn 118 Og
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
H21 P21 Since both atoms H and ...View the full answer

Answered By

Hassan Imtiaz
The following are details of my Professional Experience. Responsibilities Eight years of demanding teaching experience in the field of finance and business studies at Master’s Level. Completion of the given tasks within given time with quality and efficiency. Marketing professional with practical experience in and solid understanding of a diverse range of management applications, including market analysis, sales and marketing, team building and quality assurance. I have excellent skills to approach deal and sustain corporate clients / customers by demonstrating not only extraordinary communication and interpersonal skills but also high caliber presentation, negotiation and closing skills. Manage and follow up the day-to-day activities. Manage and co-ordinate the inventories. Fulfillment of all the tasks assigned.
The following are details of my Areas of Effectiveness. Finance 1. Corporate Finance 2. Advanced Corporate Finance 3. Management of Financial Institutions 4. International Financial Management 5. Investments 6. Fixed Income 7. Real Estate Investment 8. Entrepreneurial Finance 9. Derivatives 10. Alternative Investments 11. Portfolio Management 12. Financial Statement Analysis And Reporting (US GAAP & IFRS) 13. International Financial Markets 14. Public Finance 15. Personal finance 16. Real estate 17. Financial Planning Quantitative Analysis 1. Time Value Of Money 2. Statistics 3. Probability Distribution 4. Business Statistics 5. Statistical Theory and Methods Economics 1. Principles of Economics 2. Economic Theory 3. Microeconomic Principles 4. Macroeconomic Principles 5. International Monetary Economics 6. Money and Banking 7. Financial Economics 8. Population Economics 9. Behavioral Economics International Business 1. Ethics 2. Business Ethics 3. An introduction to business studies 4. Organization & Management 5. Legal Environment of Business 6. Information Systems in Organizations 7. Operations Management 8. Global Business Policies 9. Industrial Organization 10. Business Strategy 11. Information Management and Technology 12. Company Structure and Organizational Management Accounting & Auditing 1. Financial Accounting 2. Managerial Accounting 3. Accounting for strategy implementation 4. Financial accounting 5. Introduction to bookkeeping and accounting Marketing 1. Marketing Management 2. Professional Development Strategies 3. Business Communications 4. Business planning 5. Commerce & Technology Human resource management 1. General Management 2. Conflict management 3. Leadership 4. Organizational Leadership 5. Supply Chain Management 6. Law 7. Corporate Strategy Creative Writing 1. Analytical Reading & Writing Other Expertise 1. Risk Management 2. Entrepreneurship 3. Management science 4. Organizational behavior 5. Project management 6. Financial Analysis, Research & Companies Valuation 7. And any kind of Excel Queries
4.80+
150+ Reviews
230+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to the values in Figure 12.9 and calculate the electronegativity difference in a SI bond. Figure 12.9 1 IA S Li 1.0 11 2 IIA 4 Na 0,9 19 Be 1.5 12 0.8 37 Mg 1.2 6 20 Rb 0.8 55 Ca 1.0 38 3...
-
Refer to the values in Figure 12.9 and calculate the electronegativity difference in a SbCl bond. Figure 12.9 1 IA S Li 1.0 11 2 IIA 4 Na 0,9 19 Be 1.5 12 0.8 37 Mg 1.2 6 20 Rb 0.8 55 Ca 1.0 38 3...
-
Refer to the values in Figure 12.9 and calculate the electronegativity difference in a BCl bond. Figure 12.9 1 IA S Li 1.0 11 2 IIA 4 Na 0,9 19 Be 1.5 12 0.8 37 Mg 1.2 6 20 Rb 0.8 55 Ca 1.0 38 3...
-
Please answer all questions from 1 to 12 4. Work-Power-Energy 7. A body of mass m, accelerates uniformly from rest to v, in time t,, The instantaneous power delivered to the body as a function of...
-
1. The accounts estimated revenues and appropriations appear in the trial balance of the general fund. These accounts indicate: a. The use of cash basis accounting b. The use of accrual basis...
-
Prepare journal entries for each transaction listed. a. During the period, customer balances are written off in the amount of $17,000. b. At the end of the period, bad debt expense is estimated to be...
-
The average age of the 115 residents of a retirement community
-
Use VOTE 1.RAW for this exercise. (i) Estimate a model with voteA as the dependent variable and prtystrA, democA, log(expendA), and log(expendB) as independent variables. Obtain the OLS residuals, u,...
-
The M & L Inn's summary income statement is in the Tab 1, please use the information to answer the following questions: What is the breakeven point for the M & L Inn? (2 points) If the fixed cost...
-
Explain why the radius of a sodium ion (95 pm) is about half that of a sodium atom (186 pm).
-
Write formula units by combining the cations and anions in each of the following pairs. (a) Sc 3+ and N 3 (b) Ti 4+ and O 2 (c) NH 4+ and CO 3 2 (d) Hg 2 2+ and PO 4 3 .
-
(a) What is the temperature of the peak of a blackbody spectrum is at 18.0nm? (b) What is the wavelength at the peak of a blackbody spectrum if the body is at a temperature of 2000K?
-
Zephyr Minerals completed the following transactions involving machinery. Machine No. 1550 was purchased for cash on April 1, 2020, at an installed cost of $75,000. Its useful life was estimated to...
-
Kelly is a self-employed tax attorney whose practice primarily involves tax planning. During the year, she attended a three-day seminar regarding new changes to the tax law. She incurred the...
-
At a recently concluded Annual General Meeting (AGM) of a company, one of the shareholders remarked; historical financial statements are essential in corporate reporting, particularly for compliance...
-
4. In hypothesis, Mr. Ng wants to compare the solution in Q3 to other solutions in different conditions. If the following constraints are newly set in place, answer how much different is going to be...
-
3C2H6O2+7H2O= C2H4O3+11H2+O2+H2C2O4+CH2O2 Glycolic acid is produced electrochemically from ethylene glycol under alkaline conditions(NaOH). Hydrogen is produced at the cathode, and formic acid and...
-
A man bought a 5% tax-free municipal bond. It cost $1000 and will pay $50 interest each year for 20 years. The bond will mature at the end of the 20 years and return the original $1000. If there is...
-
QUESTION 2 The CEO of Farisha Hijab Sdn Bhd insisted on further investigation to be carried out that he also required Mr Muaz to conduct the analysis of variance for the material and labour of the...
-
Calculating Flotation Costs the Educated Horses Corporation needs to raise $40 million to finance its expansion into new markets. The company will sell new shares of equity via a general cash...
-
Calculating Flotation Costs in the previous problem, if the SEC filing fee and associated administrative expense of the offering are $900,000, how many shares need to be sold?
-
Calculating Flotation Costs the Huff Co, has just gone public. Under a firm commitment agreement, Huff received $15.05 for each of the 5 million shares sold. The initial offering price was $26 per...
-
1,600 Balance Sheet The following is a list (in random order) of KIP International Products Company's December 31, 2019, balance sheet accounts: Additional Paid-In Capital on Preferred Stock $2,000...
-
Question 3 4 pts 9 x + 3 x 9 if x 0 Find a) lim f(x), b) lim, f(x), C), lim , f(x) if they exist. 3 Edit View Insert Format Tools Table : 12pt M Paragraph B IV A2 Tv
-
Mr. Geoffrey Guo had a variety of transactions during the 2019 year. Determine the total taxable capital gains included in Mr. Guo's division B income. The transactions included: 1. On January 1,...

Study smarter with the SolutionInn App


