The chemical formulas for the oxides of potassium, calcium, gallium, and germanium are, respectively, K 2 O,
Question:
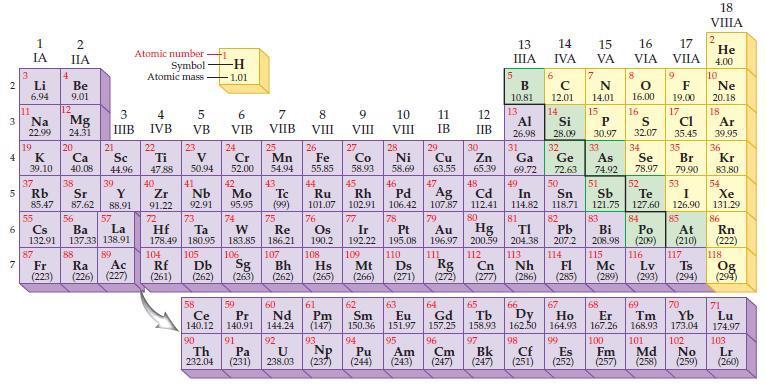
The chemical formulas for the oxides of potassium, calcium, gallium, and germanium are, respectively, K2O, CaO, Ga2O3, and GeO2. Refer to the periodic table and predict the chemical formula for each of the following compounds:
(a) Rubidium oxide
(b) Strontium oxide
(c) Indium oxide
(d) Lead oxide.
Periodic Table:
Transcribed Image Text:
2 3 4 15 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 R Rb 4 87 2 IIA Be 9.01 12 Fr (223) K Ca Sc 39.10 40.08 44.96 Mg 24.31 20 38 21 Sr Y 85.47 87.62 88.91 3 IIIB 88 39 55 56 La Cs Ba 132.91 137.33 138.91 57 89 Ra Ac (226) (227) Atomic number Symbol Atomic mass 4 IVB 22 Ti 47.88 40 Zr 2 91.22 72 5 VB 104 23 V 50.94 41 73 Hf Ta 178.49 180.95 105 Rf Db (261) (262) -H -1.01 6 VIB Nb Mo 92.91 95.95 90 24 Cr 52.00 42 74 106 Sg (263) 58 Pr Ce 140.12 140.91 91 59 7 VIIB Th Pa 232.04 (231) 25 W Re 183.85 186.21 Mn 54.94 43. Tc (99) 75 107 Bh (262) 60 Nd 144.24 92 U 238.03 8 VIII 26 Fe 55.85 76 Os 190.2 61 Pm (147) 93 Np 9 VIII (237) 10 VIII 11 IB 12 IIB 27 28 29 Zn Co Ni Cu 58.93 58.69 63.55 65.39 44 47 49 45 46 48 Ru Rh Pd Ag Cd In 101.07 102.91 106.42 107.87 112.41 114.82 77 78 80 79 Au Hg 196.97 200.59 111 112 Ir Pt 192.22 195.08 109 110 Hs Mt Ds Rg Cn (265) (266) (271) (272) (277) 108 30 5 13 ΠΙΑ B 10.81 13 6 14 IVA с 12.01 14 15 16 17 VA VIA VILA 7 N 14.01 15 66 67 62 63 64 65 Sm Eu Gd Tb Dy Ho 150.36 151.97 157.25 158.93 162.50 164.93 99 94 95 96 97 Pu Am Cm Bk (244) (243) (247) (247) 8 P 30.97 O 16.00 Al Si 31 34 53 26.98 28.09 32 33 Ga Ge As Se 69.72 72.63 74.92 78.97 50 51 52 Sn Sb Te I 118.71 121.75 127.60 126.90 82 83 84 85 TI Pb Bi Po At 204.38 207.2 208.98 (209) (210) 113 114 115 116 117 Nh Fl Mc Lv Ts (286) (285) (289) (293) (294) 81 16 9 F 19.00 17 Cl 35.45 S 32.07 35 Br 79.90 68 69 70 Er Tm Yb 167.26 168.93 173.04 100 101 102 Cf Es Fm Md No (251) (252) (257) 98 (258) (259) 18 VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39,95 36 Kr 83,80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 Lr (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a Rb 2 ...View the full answer

Answered By

Ishrat Khan
Previously, I have worked as an accounting scholar at acemyhomework, and have been tutoring busines students in various subjects, mostly accounting. More specifically I'm very knowledgeable in accounting subjects for college and university level. I have done master in commerce specialising in accounting and finance as well as other business subjects.
5.00+
134+ Reviews
426+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
The formulas for the chlorides of potassium, calcium, boron, and germanium are, respectively, KCl, CaCl 2 , BCl 3 , and GeCl 4 . Using the periodic table, predict the chemical formulas for each of...
-
The formulas for the oxides of sodium, magnesium, aluminum, and silicon are, respectively, Na 2 O, MgO, Al 2 O 3 , and SiO 2 . Using the periodic table, predict the chemical formulas for each of the...
-
Arthur Korrey is developing a project to start a new professional aquatic baseball league, with teams comprised of humans, dolphins and penguins. He forecasts that net annual cash flows will be zero...
-
Solve the inequalities in Problems 4150. 7-5A < 2A + 7
-
Comparison of Public and Private Universities. Following are the operating statements for a public and private university. The operating statements have been adapted from the annual reports of a...
-
1/Calculate the return on the ENI share and on the Italian index over 13 months until 1 January 2011. To help you, you have a record of the share price and of the general index. What is the total...
-
Figure Q28.20 shows the standing de Broglie wave of a particle in a box. a. What is the quantum number? b. Can you determine from this picture whether the "classical" particle is moving to the right...
-
Goree Company began operations on January 1, 2011, by issuing common stock for $30,000 cash. During 2011, Goree received $40,000 cash from revenue and incurred costs that required $60,000 of cash...
-
In each diagram to the right, determine (a) numbers to place in the red circles so the sum of the numbers in each pair of adjacent red circles is the number in the corresponding large blue circles....
-
Metallic sodium reacts with chlorine gas to give sodium chloride, NaCl. Predict the products formed when (a) Lithium (b) Potassium react with chlorine gas.
-
Predict the missing value (?) for each physical property listed below. The (a) Atomic radius, (b) Density, (c) Melting point are given for two of the metals in Group VIII/10. Element Ni Pd Pt Atomic...
-
For each of the events listed below, indicate whether the event represents a transactional entry, an adjusting entry, or no entry in the accounting system. Assume the accounting system is designed to...
-
List at least three reasons that demonstrate why having a business model is important.
-
What is a new-venture team? Who are the primary participants in a start-ups new-venture team?
-
What are the characteristics of a promising acquisition candidate?
-
In what ways does a focus on a cost leadership strategy lead to a very different business model than a focus on a differentiation strategy?
-
What are the advantages and disadvantages of a limited liability company? Is a limited liability company an appropriate form of ownership for an aggressive entrepreneurial firm?
-
Study the following Minitab output from a regression analysis to predict y from x. a. What is the equation of the regression model? b. What is the meaning of the coefficient of x? c. What is the...
-
(a) Explain why the concentration of dissolved oxygen in freshwater is an important indicator of the quality of the water. (b) How is the solubility of oxygen in water affected by increasing...
-
Andrews Inc., a greeting card company, had the following statements prepared as of December 31, 2010. Additional information: 1. Dividends in the amount of $6,000 were declared and paid during 2010....
-
Data for Andrews Inc. are presented in E23-13. Prepare a statement of cash flows using the indirect method.
-
Presented below are data taken from the records of Morgenstern Company. Additional information: 1. Held-to-maturity securities carried at a cost of $43,000 on December 31, 2009, were sold in 2010 for...
-
JMC Co. takes out a 3-year loan of $700,000 from a bank. It keeps $200,000 in its chequing account, and uses the rest to purchase a new sorting machine for its factory. How is shareholder's equity...
-
Given the following weighed undirected graph, determine the optimal path for the traveling salesman to travel to visit each city once. Start at city 4. Show your work using exhaustive search to get...
-
Chapter 6 - Files and Files and Exceptions Complete the following programming exercises adapted from those found at the end of Chapter 6: But note - I have made significant modifications and added...

Study smarter with the SolutionInn App


