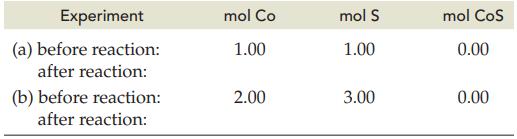
The following table indicates the amounts of Co and S before reaction. Complete the table after reactions
Question:
The following table indicates the amounts of Co and S before reaction. Complete the table after reactions (a) and (b) according to the balanced equation:
![]()
Transcribed Image Text:
Co(s) + S(s) A, CoS(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To complete the table after reactions a and b according to the balanced equati...View the full answer

Answered By

Muhammad Khurram
I have strong General Management skills to apply in your projects. Over last 3 years, I have acquired great knowledge of Accounting, Auditing, Microsoft Excel, Microsoft PowerPoint, Finance, Microsoft Project, Taxation, Strategic Management, Human Resource, Financial Planning, Business Planning, Microsoft Word, International Business, Entrepreneurship, General Management, Business Mathematics, Advertising, Marketing, Supply Chain, and E-commerce. I can guarantee professional services with accuracy.
4.80+
249+ Reviews
407+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
The following table indicates the amounts of Co and S before reaction. Complete the table after reactions (a) and (b) according to the balanced equation: 2 Co(s) + 3 S(s) C0S3(s)
-
The following partial income statement and income tax note excerpts were taken from Sirius XM Inc.'s 2012 Form 10-K. During 2012, Sirius XM eliminated most of the valuation allowance it had built up,...
-
XYZ Co is a prominent company in the FMCG industry. It provides 400 different brands spanning 14 categories of home, personal care and foods products. XYZ is now one of the worlds biggest companies....
-
Jaez Corporation is in the process of going through a reorganization. As of December 31, 2024, the companys accountant has determined the following information although the company is still several...
-
Calculate the magnitude and direction of the Coulomb force on each of the three charges shown inFigure. -2.00 C 1.50 C 6.00 uC 2.00 cm -3.00 cm
-
The profit (in thousands of dollars) that Aunt Mildreds Metalworks earns from producing x tons of steel and y tons of aluminum can be approximated by P(x, y) = 36xy - x 3 - 8y 3 . Find the amounts of...
-
(a) In Figure 7.5, what is the momentum of the ball during the collision? (b) Is the momentum of the ball constant before, during, and after the collision? If so, why? If not, why not, and for what...
-
Jem Clothes, Inc., is a 25-store chain concentrated in the northeastern United States that sells ready-to-wear clothes for young men and women. Each store has a full- time manager and an assistant...
-
Microsoft Corporation makes Xbox video game consoles. For Microsoft's financial year YYYY, please make use of the following provided information regarding the inventory of those Xbox consoles:...
-
If 50.0 g of molten iron(II) oxide reacts with 10.0 g of magnesium, what is the mass of iron produced? FeO (1) + Mg(1) Fe(l) + MgO(s)
-
If 1.00 mol of ethane gas and 3.00 mol of oxygen gas react, what is the limiting reactant and how many moles of water are produced according to the equation in Exercise 55? Exercise 55 If 1.00 mol of...
-
Acme Distributors purchases inventory in crates of merchandise. Assume the company began January with an inventory of 30 units that cost $300 each. During the month, the company purchased and sold...
-
To hedge the exposure in the previous problem, Ragnetto can alternatively use a money market hedge. Ragnetto can borrow/lend U.S. dollars at an annualized interest rate of 12% and Taiwanese dollars...
-
If an investor spends $130,000 to get a one-year bond that pays 5% nominal interest, how much will the investor receive if inflation was 6% over the course of the year in terms of real value?
-
If you buy an option that gives you the right to buy gold at $1700 an ounce in one month, but gold is now trading for $1600 an ounce, is that option valuable today? Why or why not?
-
Describe a type of healthcare organization that functions as a complex adaptive system (CAS). What are the characteristics of this organization that suggest it is a CAS? How does this differ from a...
-
Develop an operation management plan Instructions: For this activity, you will apply the concepts of operations management to real-world situations. You may wish to develop a business such as a...
-
Plant overhead for ABC Corp is $150 million per year, a portion of which (20 percent) is attributable to inspection costs which are charged to products on the basis of the number of parts in the...
-
The trade-off theory relies on the threat of financial distress. But why should a public corporation ever have to land in financial distress? According to the theory, the firm should operate at the...
-
Critique Fiedlers LPC theory. Are other elements of the situation important? Do you think Fiedlers assertion about the inflexibility of leader behavior makes sense? Why or why not?
-
Do you agree or disagree with Fiedlers assertion that leadership motivation is basically a personality trait? Why?
-
Compare and contrast the LPC and path-goal theories of leadership. What are the strengths and weaknesses of each?
-
B. Suppose one of your competitors announces that, due to increases in wages, rent, and other input prices, it is going to increase the price it charges by 10% at the start of the next month. Could...
-
Suppose the FED announces it will lower the "interest rate" several times over the next year or so (a little bit at a time). Using our macro model explain: 1. What justification would cause the FED...
-
a. Find all the pure NE. b. What NE are not sequentially rational? why? c. Apply the backward induction procedure. How many solutions do you find? 6. Consider a dynamic version of matching pennies,...

Study smarter with the SolutionInn App


