Write a balanced equation for each of the following redox reactions in an acidic solution using the
Question:
Write a balanced equation for each of the following redox reactions in an acidic solution using the half-reaction method.
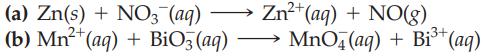
Transcribed Image Text:
(a) Zn(s) + NO3- (aq) (b) Mn²+ (aq) + BiO3(aq) Zn²+ (aq) + NO(g) MnO4 (aq) + Bi³+ (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a 3 Zns 2 NO 3 aq 8 H aq ...View the full answer

Answered By

ZIPPORAH KISIO LUNGI
I have worked on several other sites for more than five years, and I always handle clients work with due diligence and professionalism. Am versed with adequate experience in the fields mentioned above in which have delivered quality papers in research, thesis, essays, blog articles, and so forth.
I have gained extensive experience in assisting students to acquire top grades in biological, business and IT papers. Notwithstanding that, I have 7+ years of experience in corporate world software design and development.
5.00+
194+ Reviews
341+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Write a balanced equation for each of the following redox reactions in a basic solution using the half-reaction method. (a) MnO4 (aq) + S(aq) (b) Cu(s) + CIO (aq) MnO(s) + S(s) Cu+ (aq) + Cl (aq)
-
Write a balanced equation for each of the following reactions or reaction sequences. (a) The reaction catalyzed by PFK-2 (b) The conversion of 2 moles of oxaloacetate to glucose (c) The conversion of...
-
Write a balanced equation for each of the following redox reactions using the oxidation number method. (a) Fe 2+ (aq) + H 2 O 2 (aq) + H + (aq) Fe 3+ (aq) + H 2 O(l) (b) Cr 2 O 7 2 (aq) + Br (aq) +...
-
In recent years, Avery Transportation purchased three used buses. Because of frequent turnover in the accounting department, a different accountant was in charge of selecting the depreciation method...
-
Prepare journal entries to record these transactions: (a) Benton Company retires its delivery equipment, which cost $41,000. Accumulated depreciation is also $41,000 on this delivery equipment. No...
-
Find the reference angle and the exact function value if they exist. cot ( -90)
-
A current-carrying wire is bent into a circular loop of radius \(R\) and lies in an \(x y\) plane. A uniform external magnetic field in the \(+z\) direction exists throughout the plane of the loop....
-
Analysis of transactions and preparation of the income statement and balance sheet Zealock Bookstore opened a bookstore near a college campus on July 1, 2008. Transactions and events of Zealock...
-
19. Find the tension T for the system shown in figure :- T T T 1 kg 2 kg 3 kg (1) IgN (2) 2 gN (3) 5 gN (4) 6 gN 20. A ball of mass 0.5 kg moving with a velocity of 2 m/sec strikes a wall normally...
-
Write a balanced half-reaction for each of the following in a basic solution. (a) Ni(OH) 2 (s) NiO 2 (s) (b) NO 2 (aq) N 2 O(g).
-
Write a balanced half-reaction for each of the following in an acidic solution. (a) H 2 O 2 (aq) H 2 O(l) (b) AsO 3 3 (aq) AsO 3 (aq).
-
You will be given $50 per day for meals when you travel, so you no longer need to provide receipts. Identify which job design technique is exemplified in each statement. A. Job simplification B. Job...
-
Supply chain analysis is part of value chain analysis. This news item indicates the types of businesses that feel they need to know more about their supply chains. Six leading consumer products...
-
What have researchers found about the frequency of review of working capital in small businesses.
-
What is the just-in-time approach to purchasing inventory.
-
The following table sets out information in respect of Division X and Division Y. The cost of borrowing new finance is 10% per annum. Required Explain what view the managers of each division might...
-
As technology becomes more sophisticated and complicated, which challenges presented by virtual teams might become more problematic?
-
Define the term internal rate of return or IRR. Describe how you would calculate the IRR for a project proposal.
-
A report from the college dean indicates that for the previous semester, the grade distribution for the Department of Psychology included 135 As, 158 Bs, 140 Cs, 94 Ds, and 53 Fs. Determine what kind...
-
The following accounts and their balances were selected from the unadjusted trial balance of REO Inc., a freight forwarder, at October 31, the end of the current fiscal year: Preferred 2% Stock, $100...
-
The following accounts and their balances appear in the ledger of Newberry Properties Inc. on June 30 of the current year: Common Stock , $75 par $1,350,000 Paid-In Capital in Excess of Par 108,000...
-
Race Car Inc. retails racing products for BMWs, Porsches, and Ferraris. The following accounts and their balances appear in the ledger of Race Car Inc. on April 30, the end of the current year:...
-
Fast Iguana had a great year. It grew its revenues by 20% while maintaining its Gross Profit Margin (GPM) at 22%. Fast Iguana reported Net Income of $95M. You are the CFO of Fast Iguana and your...
-
Discuss the relevance of (QBO) QuickBooks online, and how this course will help you in your career. What are your thoughts on how (QBO) QuickBooks Online can be an impactful resource in helping an...
-
Techsphere Enterprises is a west coast manufacturing company that uses a process costing system. At the start of production, Techsphere adds all the raw material that will be need for production....
Blood Money Compilation Of Articles By Anna Von Reitz 1st Edition - ISBN: 979-8453405404 - Free Book

Study smarter with the SolutionInn App


