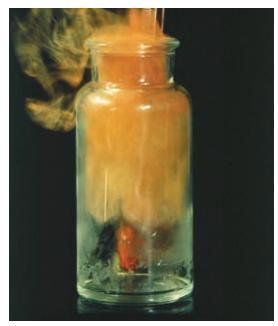
Write a chemical equation for iron metal combining with chlorine gas to give yellow ferrous chloride solid.
Question:
Write a chemical equation for iron metal combining with chlorine gas to give yellow ferrous chloride solid.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The chemical equation for the reaction described is 2 Fes 3 Cl2g 2 FeCl3s Explanation I...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
The reaction of iron metal and chlorine gas to give iron(III) chloride is illustrated below. (a) Write the balanced chemical equation for the reaction. (b) Beginning with 10.0 g of iron, what mass of...
-
Write a chemical equation for magnesium metal combining with bromine liquid to give solid magnesium bromide.
-
In each of the following pairs of compounds choose which one will react faster with the indicated reagent, and write a chemical equation for the faster reaction: (a) Toluene or chlorobenzene with a...
-
Wical Rental Management Services manages four apartment buildings, each with a different owner. Wicals CEO has observed that the apartment buildings with more expensive rental rates tend to require...
-
The ledger of Conway Corporation at December 31, 2012, after the books have been closed, contains the following stockholders equity accounts. Preferred Stock (10,000 shares issued)................
-
Elizabeth Greene wants to buy 300 shares of Google, which is selling in the market for $537.34 a share. Rather than liquidate all her savings, she decides to borrow through her broker at 5 percent a...
-
A fellow scientist heard that a Van de Graaff generator built 70 years ago could collect \(5.0 \mathrm{C}\) of charge on its dome, which had a radius of \(1.1 \mathrm{~m}\), and has challenged you to...
-
Sylvan Inc. entered into a non-cancelable lease arrangement with Breton Leasing Corporation for a certain machine. Bretons primary business is leasing; it is not a manufacturer or dealer. Sylvan will...
-
The _________ provides a portion of the system call interface for UNIX and Linux.
-
Write a chemical equation for the reaction of cadmium metal with aqueous cobalt(II) nitrate to produce aqueous cadmium nitrate and cobalt metal.
-
Write a balanced chemical equation for each of the following combination reactions: (a) Calcium metal is heated with fluorine gas to yield solid calcium fluoride. (b) Manganese metal reacts with...
-
1. What does senior management want? 2. What does Brenda want? 3. What does the Florida sales team want? 4. How would you describe the Florida sales team? 5. Describe the systems at play here. 6. If...
-
Financial advisor Joseph Laterreur recommends to his client Fatima, who is willing to assume relatively high risk, the following investment strategy regarding the stock of Moderna Inc. (ticker:...
-
Analyze the impact of leverage on the risk and return of an investment portfolio. Consider margin trading and its potential consequences.
-
9) Lonnie can save 15% on his insurance if he pays it within 15 days of receipt. If he pays $446.25 on the 10th day, what is the original amount of his bill? 10) 24% of what number is 60? 11) A high...
-
KUALA LUMPUR: The FBM KLCI hastened its decline headed into midday as the blue-chip counters fell in unison on the Budget 2022 proposal of a one-off special tax on high-income companies. At 12.30pm,...
-
Discuss how a company can improve its production and sales
-
List benchmarks or criteria often used in assessing productivity, and discuss their advantages and disadvantages.
-
Show that gj concave AHUCQ Abadie For nonnegative variables, we have the following corollary.
-
In 2010 Ghostbusters Corp. spent $420,000 for goodwill visits by sales personnel to key customers. The purpose of these visits was to build a solid, friendly relationship for the future and to gain...
-
What are factors to be considered in estimating the useful life of an intangible asset?
-
What should be the pattern of amortization for a limited life intangible?
-
2.1 Design and determine data storage requirements from NoSQL data store according to selected vendor technology and business requirements
-
Determine profit from the following transaction. You purchase 55 SPI200 futures contracts at 7372 and hold them to expiry when the market settles at 7322.50. In your answer you must state your...
-
28. Below is the model of a three-dimensional structure. Draw the front view of this model. Each cube is exactly inch in length, width, and height. Use a scale of 2:1. (2 marks) Back Top Front Left...

Study smarter with the SolutionInn App


