Write the solubility product expression for each of the following slightly soluble ionic compounds in a saturated
Question:
Write the solubility product expression for each of the following slightly soluble ionic compounds in a saturated aqueous solution:
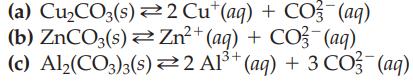
Transcribed Image Text:
(a) Cu₂CO3(s) 2 Cut(aq) + CO2 (aq) (b) ZnCO3(s)Zn²+ (aq) + CO3(aq) (c) Al₂(CO3)3(s) 2 Al³+ (aq) + 3CO3(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The solubility product expression for a slightly soluble ionic compound in a saturated aqueous s...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Write the solubility product expression for each of the following slightly soluble ionic compounds in a saturated aqueous solution: (a) Agl(s) Ag+ (aq) + (aq) (b) AgCrO4(s) (c) Ag3PO4(s) 2 Ag+ (aq) +...
-
When a pure substance is placed in contact with water, there are three possible outcomes. The substance may do nothing that is, the substance does not dissolve and no visible change takes place. The...
-
Write the solubility product expression for slightly soluble aluminum carbonate in an aqueous solution. Al 2 (CO 3 ) 3 (s) 2 Al 3+ (aq) + 3 CO 3 2 (aq)
-
What is the future value of the following cash flows, given an appropriate discount rate of 7.74% (to the nearest penny)? Year 1 Year 2 Year 3 Year 4 Year 5 $2,005 $4,054 $6,159 $8,532 $11,985
-
Assume that interest rate parity holds. In both the spot market and the 90-day forward market, 1 Japanese yen equals 0.0086 dollar. In Japan, 90-day risk-free securities yield 4.6%. What is the yield...
-
A function f is defined as for x 0. Find an expression for f(x) and determine whether f is an increasing function, a decreasing function or neither. f(x) = 5 (x + 22 2 x + 2
-
Dr. Wayside works at a local clinic as a general practitioner. Because she has significant unsecured debt, Dr. Wayside decided to petition the United States Bankruptcy Court for relief under Chapter...
-
Rachel wants to determine the shipping routes that will minimize Statelines total cost in order to develop a contract proposal to submit to Polychem for waste disposal. She particularly wants to know...
-
10. A plastic ball is rising in water with terminal speed v. If we use a different plastic whose density is double that of the original ball but still less that density of water, keeping the radius...
-
Given the chemical equation for the ionization of ammonium hydroxide NH 4 OH(aq) NH 4 +(aq) + OH (aq) Predict the direction of equilibrium shift for each of the following stresses: (a) Increase [NH...
-
Given the chemical equation for the ionization of acetic acid HC 2 H 3 O 2 (aq) H + (aq) + C 2 H 3 O 2 (aq) Predict the direction of equilibrium shift for each of the following stresses: (a)...
-
In October 2009, Matthew bought a piece of rare porcelain for 10,000. The porcelain was damaged in early 2016 and in February of that year Matthew spent 3,850 on restoration work. In July 2016,...
-
True Or False Specialists are held to a higher standard of care than other members of the profession.
-
What is a reasonable person expected to know?
-
How is breach of duty determined?
-
What is the voluntary-undertaking doctrine?
-
Who is considered an invitee, and what duty of care is owed an invitee? a. How can one lose ones invitee status?
-
Melissa Mertz, president of Mertz Industries, asked controller Rick Caputo to prepare a report on the use of electricity by each of the organizations five divisions. Increases in electricity costs in...
-
C- Consider the following scenario:- A supermarket needs to develop the following software to encourage regular customers. For this, the customer needs to supply his/her residence address, telephone...
-
Green Thumb Gardening is a small gardening service that uses activity-based costing to estimate costs for pricing and other purposes. The proprietor of the company believes that costs are driven...
-
Klumper Corporation is a diversified manufacturer of industrial goods. The companys activity- based costing system contains the following six activity cost pools and activity rates: Activity data...
-
Thermal Rising, Inc., makes paragliders for sale through specialty sporting goods stores. The company has a standard paraglider model, but also makes custom-designed paragliders. Management has...
-
Consumers are paying record amounts of fees for overdrawing their bank accounts. In 2007, banks, thrifts, and credit unions collected $45.1 billion in overdraft fees, which is $14.1 billion more than...
-
When I first started practice in 1987, the top individual income tax rate was 38.5%, and the top corporate tax rate was 34.6%. At that time, whether shareholder salaries in a closely held corporation...
-
5. Imagine that the following Treasury bonds are available: Maturity 1 Coupon Rate 0% Price $ 94.340 9.5% 0% $105.403 $ 80.496 3 3 Maturity is in years, coupons are paid annually, and prices are...

Study smarter with the SolutionInn App


