At a fixed temperature, the volume of a sample of gas varies inversely as the pressure of
Question:
At a fixed temperature, the volume of a sample of gas varies inversely as the pressure of the gas. If the pressure of a sample of gas at a fixed temperature is increased by 50%, by what percent is the volume decreased?
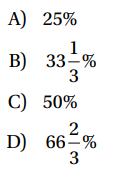
Transcribed Image Text:
A) 25% 1 33-% B) 3 C) D) 50% 2 66-% 3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
B Pick values for the original pressure and vo...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

McGraw-Hill Education SAT 2017
ISBN: 9781259641657
1st Edition
Authors: Christopher Black, Mark Anestis
Question Posted:
Students also viewed these Business questions
-
Solve each problem The volume of a gas varies inversely as the pressure and directly as the temperature in kelvins (K). If a certain gas occupies a volume of 1.3 L at 300 K and a pressure of 18...
-
The associate administrator at Getwell Hospital wants to evaluate the layout of the outpatient clinic. Table 10.7 shows the interdepartmental flows (patients/day) between departments; Figure 10.15...
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
Complete the questions in the table below.
-
Mrs. Gordon Neely hired Louise Bradshaw as the bookkeeper for a Midas Muffler shop they owned and operated as a corporation, J. Gordon Neely Enterprises, Inc. (Neely). Bradshaw's duties included...
-
Explain how the center of gravity method works.
-
What the general requirements are for all pleadings?
-
James Hardy recently rejected a $20,000,000, five-year contract with the Vancouver Seals. The contract offer called for an immediate signing bonus of $5,000,000 and annual payments of $3,000,000. To...
-
How can we win the Fed the Chair game? Need a summary report on the simulation when objective is to keep inflation near 2% and unemployment near 5%, using the fed funds rate
-
Name the fallacy in each statement below and explain why it is fallacious. 1. The best restaurant in New York city is either buddy's bistro or clyde's emporium. 2. Everyone is going to buddy's...
-
Between 1970 and 2010, the annual consumption of fossil fuels in the U.S. increased nearly linearly. If this linear trend were to continue, which of the following is closest to the level of U.S....
-
If f(x) = x 2 + x + k, where k is a constant, and f(2) = 10, what is the value of f(-2)?
-
Put in order of increasing rate of substitution by H 2 O the complexes (a) [Co(NH 3 ) 6 ] 3+ , (b) [Rh(NH 3 ) 6 ] 3+ , (c) [Ir(NH 3 ) 6 ] 3+ , (d) [Mn(OH 2 ) 6 ] 2+ , (e) [Ni(OH 2 ) 6 ] 2+ .
-
Your Client Mr. Smith has booked the appointment with your associate and is exploring a variety of Investment and retirement planning options. He wants to better understand the offerings available to...
-
Q1. Write down the expression for the Weizsacker semi-emperical Mass Formula, explaining the reasoning behind each of the following terms a) The Volume term b) The Surface term c) The Coulomb Term d)...
-
Catherine has worked for a company in Edmonton, Alberta since 2 0 1 2 . She earns $ 3 7 . 3 5 per hour, works 4 0 hours per pay period and is paid on a weekly basis. The company provides its...
-
For each of the following independent cases (A to E), compute the missing values in the table: Case Prime Cost Conversion Cost Direct Materials Direct Manufacturing Labor Overhead Total Manufacturing...
-
On June 29, Year 3, Sullivan purchased and placed into service an apartment building costing $360,000 including $30,000 for the land. What was Sullivan's MACRS deduction for the apartment building in...
-
State the differences between rules and norms and how norms are enforced.
-
From a medical tourist perspective, compare Shouldice with the traditional hospital in terms of the key factors of competition. Using Table 15-3, why would Shouldice attract patients from outside the...
-
Daniel receives 400 shares of A&M Corporation stock from his aunt on May 20, 2017, as a gift when the stock has a $60,000 FMV. His aunt purchased the stock in 2007 for $42,000. The taxable gift is...
-
Tally owns a house that she has been living in for eight years. She purchased the house for $245,000 and the FMV today is $200,000. She is moving into her friends house and has decided to convert her...
-
Kathleen owns 500 shares of Buda Corporation common stock which was purchased on March 20, 1999, for $48,000. On October 10 of the current year, she receives a distribution of 500 stock rights. Each...
-
A planet of mass 8.751024 kg is at location ( 5.50x1011, -2.75x1011, 0) m. A star of mass 5.001030 kg is at location < -4.501011, 6.001011, 0> m. It will be useful to draw a diagram of the situation,...
-
Consider the pressure - volume diagram shown with the following values: p 1 = 2 . 4 atm p 2 = 5 . 9 atm IN 1 = 2 . 4 L IN 2 = 8 . 6 L During the process labeled A B , 2 4 0 0 J of heat is added to...
-
You are lying in your bedroom, resting after doing your physics homework. As you stare at your ceiling, you come up with the idea for a new game. You grab a dart with a sticky nose and a mass of 18.0...

Study smarter with the SolutionInn App


