In 1942, Boris Podolsky proposed a generalization of electrostatics that eliminates the divergence of the Coulomb field
Question:
In 1942, Boris Podolsky proposed a generalization of electrostatics that eliminates the divergence of the Coulomb field for a point charge. His theory retains ∇ ×E = 0 but replaces Gauss’ law by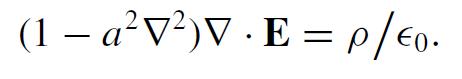
(a) Find the electric field predicted by this equation for a point charge at the origin by writing E = −∇ϕ and integrating the equation over an infinitesimal spherical volume. It will be convenient at some point to write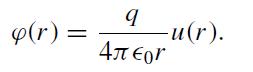
(b) Suggest a physical meaning for the parameter a.
Transcribed Image Text:
(1 − a²V²)V. E = p/ €0.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
a Because E 0 we can still define a scalar potential E Substituting this into the Podol...View the full answer

Answered By

Akshay Agarwal
I am a Post-Graduate with a specialization in Finance. I have been working in the Consulting industry for the past 8 years with a focus on the Corporate and Investment Banking domain. Additionally, I have been involved in supporting student across the globe in their academic assignments and always strive to provide high quality support in a timely manner. My notable achievements in the academic field includes serving more than 10,000 clients across geographies on various courses including Accountancy, Finance, Management among other subjects. I always strive to serve my clients in the best possible way ensuring high quality and well explained solutions, which ensures high grades for the students along-with ensuring complete understanding of the subject matter for them. Further, I also believe in making myself available to the students for any follow-ups and ensures complete support and cooperation throughout the project cycle. My passion in the academic field coupled with my educational qualification and industry experience has proved to be instrumental in my success and has helped me stand out of the rest. Looking forward to have a fruitful experience and a cordial working relationship.
5.00+
179+ Reviews
294+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Physical theory at the time Rutherford proposed his nuclear model of the atom was not able to explain how this model could give a stable atom. Explain the nature of this difficulty.
-
A positive point charge q1 creates an electric field of magnitude E1 at a spot located at a distance r1 from the charge. The charge is replaced by another positive point charge q2, which creates a...
-
Point charge q1 = -5.00 NC is at the origin and point charge q2 = +3.00 NC is on the x-axis at x = 3.00 cm. Point P is on the y-axis at Y = 4.00 cm. (a) Calculate the electric fields E1 and E2 at...
-
Table 2-5 Estonia Finland One Cell Phone 40 hours 16 hours Lumber (per board foot) 8 hours 4 hours Table 2-5 shows the number of labor hours required to produce a cell phone and a board foot of...
-
The OECD (Organization for Economic Cooperation and Development) consists of advanced, industrialized countries that accept the principles of representative democracy and a free market economy. For...
-
Give the reason why a dielectric material increases capacitance compared with what it would be with air between the plates of a capacitor. What is the independent reason that a dielectric material...
-
Identify treatments and experimental units in the following scenarios. a. City A would like to evaluate whether a new employment training program for the unemployed is more effective compared to the...
-
Can Executive Aviation establish an artisans lien on the plane? Why or why not? Air Ruidoso, Ltd., operated a commuter airline and air charter service between Ruidoso, New Mexico, and airports in...
-
12. The following comparison tree sorts the four values a, b, c, and d. c:d b:d b:d c:d b:d a:d a:d b:c a:d a:d c:d a:d
-
The Ploughman family has owned and operated a 640-acre farm for several generations. The family now needs to make a decision about the mix of livestock and crops for the coming year. By assuming that...
-
If the photon had a mass m, Gauss law with E = changes from 2 = / 0 to an equation which includes a lenght L = /mc: Experimental searches for m use a geometry first employed by Cavendish where a...
-
A surface current density K(r S , t) flows in the z = 0 plane which separates region 1 (z > 0) from region 2 (z < 0). Each region contains arbitrary, time-dependent distributions of charge and...
-
Consider the medical diagnosis of breast cancer with mammograms. An AP story (September 19, 2002) said that a woman has about a 50% chance of having a false-positive diagnosis over the course of 10...
-
At the beginning of January of the current year, Sorrel Co.'s ledger reflected a normal balance of $72,000 for accounts receivable. During January, the company collected $18,800 from customers on...
-
3. Convert the following decimal numbers to 8-bit binary (a) sign magnitude and (b) twos complement numbers. You must show all of your work in deriving your answer. i. -34 ii. -45 +23 iv. -62
-
According to current campaign finance laws, which of these can a super PAC do to influence elections? Donate directly to a candidate's campaign. Strategize directly with a candidate. Pay for...
-
1. If the Internet and other technologies reduce transaction costs (such as ordering costs) to zero, what effect might this have on the economic order quantity (EOQ) calculations discussed in Chapter...
-
How do people plan to navigate your career as an HSPP in which you are a highly equipped generalist qualified to do many different jobs within the human services field?
-
The data in Exercise 4 were generated using the following functions. Use the cubic splines constructed in Exercise 4 for the given value of x to approximate f (x) and f'(x), and calculate the actual...
-
Prove the following D,(cos x) = - sin x (Hint: Apply the identity cos(A + B) = cos A cos B sin A sin B)
-
Which will be more soluble (moles of metal dissolved per liter of solution), Ba(IO 3 ) 2 (K sp = 1.5 10 -9 ) or Ca(IO 3 ) 2 (K sp = 7.1 10 -7 )? Give an example of a chemical reaction that might...
-
Fe(III) precipitates from acidic solution by addition of OH - to form Fe(OH) 3 (s). At what concentration of OH - will [Fe(III)] be reduced to 1.0 10 -10 M? If Fe(II) is used instead, what...
-
Is it possible to precipitate 99.0% of 0.010 M Ce 3+ by adding oxalate (C 2 O 4 2- ) without precipitating 0.010 M Ca 2+ ?
-
Question 2 (a) Discuss the reliability challenges for electro-mechanical drive trains in offshore wind and marine energy applications. Give qualitative examples comparing different machine types in...
-
High blood Pressure: Keeping a Healthy Heart What is high blood pressure? What puts someone at risk of high blood pressure? What high blood pressure may cause?
-
Define and explain the concepts: Job Satisfaction Job Dissatisfaction You are also required to give relevant examples of three (3) cases of Job Satisfaction and three (3) cases of Job...

Study smarter with the SolutionInn App


