The text proved that the force on an isolated dielectric is Where E(r) is the total field
Question:
The text proved that the force on an isolated dielectric is 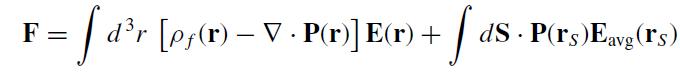
Where E(r) is the total field at an interior point r and Eavg(rS) is the average of the total field just inside and just outside the dielectric at the surface point rS. Show that this expression can be rewritten in the form: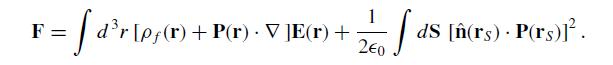
Transcribed Image Text:
F: || = · [ d³r [Pƒ(r) — ▼ · P(r)] E(r) + - . + fas ds. P(rs)Eavg (rs)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
We set f 0 because this piece requires no comment Otherwise we use Now the matching con...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Show that Eq. (3.70) can be rewritten as where C = 4Ï 2 c 2 ϵ 0 m e /Nq 2 e . |(1 1)1 = -Cx? + CAq
-
Show that the electric field just outside the surface of a good conductor of any shape is given by E = /0, where is the charge per unit area on the conductor. [Hint: The electric field...
-
Show that an isolated photon cannot be converted into an electron-positron pair, y e + e. (The conservation laws allow this to happen only near another object).
-
On January 2, 2013, Parker Corporation invests in the stock of Quarry Corporation. Quarry's book value is $4 million and its assets and liabilities are fairly reported. Quarry reports income of $3...
-
Osprey Corporation stock is owned by Pedro and Pittro, who are unrelated. Pedro owns 50% and Pittro owns 50% of the stock in the corporation. Osprey has the following assets (none of which were...
-
In a mitochondrion, where are the electron transport systems and enzymes required for ATP formation located?
-
Discuss what steps can be taken in the healthcare setting to help prevent the spread of AIDS.
-
Jack Herron is an IT auditor with McGee LLP, a large national public accounting firm. His manager, Amanda McDermott, has assigned him to the Linz Company audit. The McGee financial auditors have...
-
You want to buy a piece of farm equipment for $70,000 and the finance office has quoted you a loan with an APR of 12% compounded monthly for 60 months to make the purchase. What will your monthly...
-
A compound has a molecular formula of C8H14O4, and its infrared spectrum contains an intense peak at 1730 cm -1. The 1H NMR spectrum of the compound is shown in Figure 20.11. What is its structure?...
-
Point charges q 1 , q 2 , . . ., q N are embedded in a body with permittivity in . The latter is itself embedded in a body with permittivity out . Find the total polarization charge Q pol induced...
-
Write the Helmholtz theorem expression for D(r) and eliminate D itself from the integrals you write down. How does this formula simplify (if at all) for simple dielectric matter?
-
Plot each point in a rectangular coordinate system. (3, -3)
-
A sports memorabilia collector propositions a sport journalist with access to the locker rooms during Super Bowl LI to infiltrate the New England Patriots locker room and take Tom Brady's jersey in...
-
Cocaine inoculations: Imagine that a vaccine that would "teach" the body's immune system to attack cocaine molecules once they were introduced into the body could be developed. The development of...
-
Consider the objective function to maximize profit: Max 2A + B S.T.: Constraint 1 A + B = 1.5 non-negativity A,B >= 0 Assume the optimal solution is A-3.5 and B-0. Which of the following is FALSE? If...
-
Explain the importance and legal significance of primary business entities: Compare advantages and disadvantages of corporations and the laws and legal risks relating to this type of business entity.
-
Why is sexual harassment morally wrong? Why should sexual harassment be eliminated and legally prohibited? Explain briefly
-
What do you think should be done to revitalize the Gap store brand over the next three years?
-
Juanita owns a home in Richardson, TX. She purchases a Homeowners Policy (HO-3) from Farm State Ins. Co. The policy provides $100,000 in liability coverage (coverage E) and $5,000 in Med Pay coverage...
-
Cresol red has two transition ranges listed in Table 10-3. What color would you expect it to be at the following pH values? (a) 0; (b) 1; (c) 6; (d) 9 Table 10-3 Acid color Transition Base Indicator...
-
Would the indicator bromocresol green, with a transition range of pH 3.85.4, ever be useful in the titration of a weak acid with a strong base?
-
(a) What is the pH at the equivalence point when 0.030 0 M NaF is titrated with 0.060 0 M HClO 4 ? (b) Why would an indicator end point probably not be useful in this titration?
-
1. From the HR director: Can you let me have any research evidence that indicates what the most successful approach to recruiting graduates, or combination of approaches, is likely to be? 2. From the...
-
Using state diagram, analyze the following sequential circuit: 11 Q Clk Ck FF D 2 Q2 Ck FF K Q
-
A mixture of 52.4nM analyte X and 38.9 nM standard (S) gave the relative response Ax = 0.644 and As = 1.00. A second solution containing an unknown amount of X and 742 nM standard S had areas of Ax...

Study smarter with the SolutionInn App


