Question: Given a 0 = 1, a 1 = 5, a 2 = 2, a 3 = 7, a 4 = 1 and b 0 =
Given a0 = 1, a1 = 5, a2 = 2, a3 = 7, a4 = –1 and b0 = 0, b1 = 2, b2 = –2, b3 = 11, b4 = 3, calculate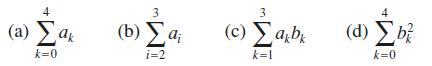
03 (2) k=0 3 ) j=2 3 ygo 3 (0) I=y A (d) b k=0
Step by Step Solution
3.45 Rating (152 Votes )
There are 3 Steps involved in it
a b c d 4 a a a a a ... View full answer

Get step-by-step solutions from verified subject matter experts


