A sample of HCl is illuminated with light of wavelength 435.8 nm. (a) Compute the wavelengths of
Question:
A sample of HCl is illuminated with light of wavelength 435.8 nm.
(a) Compute the wavelengths of the four lines in the rotational Raman spectrum that are closest to that of the incident light.
(b) Compare the difference in their frequencies with the corresponding lines in Figure 9-30.
Figure 9-30
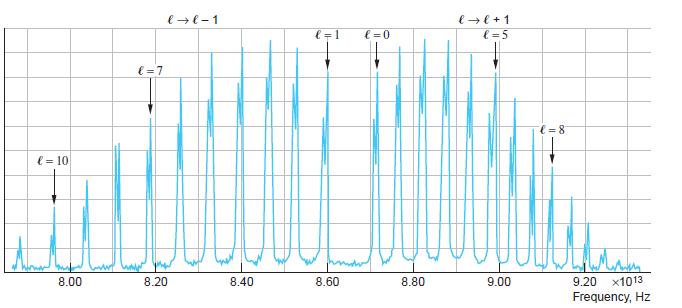
Transcribed Image Text:
l=10 8.00 €=7 8.20 l → l-1 8.40 l=1 8.60 l=0 8.80 l → l + 1 l=5 9.00 € = 8₁ 9.20 x1013 Frequency, Hz
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
a b From Figure 929 f 01 0610 Hz and f 12 12 10 Hz The agreement is ver...View the full answer

Answered By

CHARLES AMBILA
I am an experienced tutor with more than 7 years of experience. I have helped thousands of students pursue their academic goals. My primary objective as a tutor is to ensure that students have easy time handling their academic tasks.
5.00+
109+ Reviews
323+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
A slit of width 0.50 mm is illuminated with light of wavelength 500 nm, and a screen is placed 120 cm in front of the slit. Find the widths of the first and second maxima on each side of the central...
-
When a surface is illuminated with light of wavelength 512 nm, the maximum kinetic energy of the emitted electrons is 0.54eV. What is the maximum kinetic energy if the surface is illuminated with...
-
When a double-slit setup is illuminated with light of wavelength 632.8 nm, the distance between the center of the central bright position and the second side dark position is 4.5 cm on a screen that...
-
Is the PS assessment a valid predictor of performance as a store manager? Would you recommend the PS be used in the future to select sales people for promotion to store manager?
-
Ottis, Inc., uses 640,000 plastic housing units each year in its production of paper shredders. The cost of placing an order is $30. The cost of holding one unit of inventory for one year is $15.00....
-
Podrive Company manufactures computer hard drives. The market for hard drives is very competitive. The cur- rent market price for a computer hard drive is $45. Podrive would like a profit of $15 per...
-
Colin Agee, chairperson of the board of directors and chief executive officer of Image, Inc., is pondering a recommendation to make to the firm's board of directors in response to actions taken by...
-
Presented below and on the next page is the balance sheet of Rasheed Brothers Corporation (000s omitted). InstructionsEvaluate the balance sheet presented. State briefly the proper treatment of any...
-
Rachel receives employer-provided health insurance. The employer's cost of the health insurance is $6900 annually. What is her employer's after-tax cost of providing the health insurance, assuming...
-
You are a financial advisor working in Scarborough, Ontario. One day, you get a phone call from Priya Patel and you arrange a first meeting with her. You find out that she is a referral from one of...
-
The five lowest levels of a certain monatomic gas have the values E 1 = 0, E 2 = 3.80 eV, E 3 = 4.30 eV, E 4 = 7.2 eV, and E 5 = 7.5 eV. (a) If the temperature is high enough that all levels are...
-
For NaCl compute (a) The energy in eV necessary to excite the first rotational state and (b) The wavelength and frequency of the photon emitted in the transition back to the ground state. (Assume...
-
Jesse is a resident of New Jersey who works in New York City. He also owns rental property in South Carolina. During 2020, he pays the following taxes: New Jersey state estimated tax payments...
-
a) As a currency trader, you take note of the following foreign exchange rates and interest rates quotes. There are several different exchange rate circumstances for currency transaction. Spot rate...
-
public class A { int funcl (final int x) { x = x*j; return x; 13 Why is there an error here? 15 16 } 17 }
-
Compute the correlation matrix for the following data. X=[1,2,3] Y=[4,5,6] Also, write pseudo code for Correlation Based Feature Selection.
-
Explain how demand theory is reflected in the Memorial Health Systems organizational financial statements. Explain how and why market behavior affects the financial statements at various times...
-
Choose any one of the articles from the link below ... read it and in this forum review it for your classmates. Summarize the key points and your views of them. You must use the terms and knowledge...
-
Give production rules in extended Backus-Naur form that generate all decimal numerals consisting of an optional sign, a nonnegative integer, and a decimal fraction that is either the empty string or...
-
A genetically engineered strain of Escherichia coli (E. coli) is used to synthesize human insulin for people suffering from type I diabetes mellitus. In the following simplified reaction scheme,...
-
Motor neurons in mammals transmit signals from the brain to skeletal muscles at approximately 25 m/s. Estimate how long in ms it takes a signal to get from your brain to your hand?
-
Motor neurons in mammals transmit signals from the brain to skeletal muscles at approximately 25 m/s. Estimate how long in ms it takes a signal to get from your brain to your hand?
-
A Porsche accelerates from a stoplight at 5.0 m/s 2 for five seconds, then coasts for three more seconds. How far has it traveled? Draw a complete pictorial representation. Do not solve these...
-
You are considering a stock investment in one of two firms (LotsofDebt, Incorporated and LotsofEquity, Incorporated), both of which operate in the same industry. LotsofDebt, Incorporated finances its...
-
Blooper Industries must replace its magnoosium purification system. Quick & Dirty Systems sells a relatively cheap purification system for $16 million. The system will last 4 years. Do-It-Right sells...
-
3 2 points Problem 7-1 (LO7.1) Based on the following data, calculate the items requested: Rental Costs Annual rent Buying Costs eBook Hint Print References $ 8,080 Annual mortgage payments Insurance...

Study smarter with the SolutionInn App


