Assign the bands in the benzene spectra displayed in Figures 10.12 and 10.13. Figure 10.12 Figure 10.13
Question:
Assign the bands in the benzene spectra displayed in Figures 10.12 and 10.13.
Figure 10.12
Figure 10.13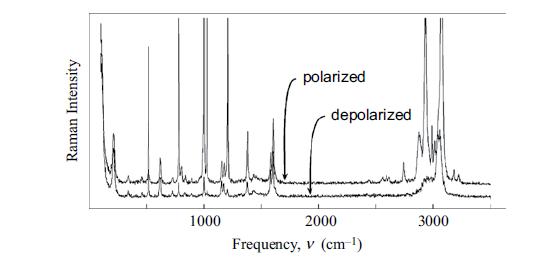
Transcribed Image Text:
Absorbance benzene Qu m. 2000 Frequency, v (cm-¹) toluene 1000 3000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (6 reviews)
The provided images show the spectra of benzene indicating various absorption and Raman intensity ba...View the full answer

Answered By

Geoffrey Isaboke
I am an industrious tutor with a 5-yr experience in professional academic writing. I have passion for History and Music and I have good knowledge in Economics
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The infrared spectra for three compounds are provided. Each compound has one or more of the following functional groups: conjugated ketone, ester, amide, nitrile, and alkyne. Determine the functional...
-
Spectra are given for three compounds. Each compound has one or more of the following functional groups: alcohol, amine, ketone, aldehyde, and carboxylic acid. Determine the functional group(s) in...
-
The following spectra are taken from a compound that is an important starting material for organic synthesis. Determine the structure, first by considering each spectrum individually, then by...
-
Adam Hoover bought 72 shares of General Oil Co. stock at a par value of $85 per share. The stock paid annual dividends of 7 1/2%. How much did Adam receive in dividends this year? Commissions 0. Mrs....
-
Ryan Inc. had the following condensed balance sheet at the end of operations for 2013. During 2014 the following occurred. 1. Land was purchased for $29,000. 2. Notes payable in the amount of $45,500...
-
Below are the last three years financial statements of Sentec Inc., a distributor of electrical fixtures. a. Compute Sentec Inc.s working capital requirement (WCR) and prepare its managerial balance...
-
A parallel-plate capacitor carries a charge \(+q\) on one plate and a charge \(-q\) on the other plate. Each plate has an area \(A\). How much force, in terms of \(q\) and \(A\), does one plate exert...
-
Parkland Department Store is located near the Lyndale Shopping Mall. At the end of the companys fiscal year on December 31, 2012, the following accounts appeared in its adjusted trial balance....
-
6. If the tangent to the curve y = x at the point P(t, t) meets the curve again at Q, then the ordinate of the point which divides PQ internally in the ratio 1:2 is: (a) 0 7. (b)-t The system of...
-
The absorption spectrum of I 2 has E 00 /hc = 15,677 cm 1 , and the onset of the continuum is at 19,735 cm 1 . The excited state dissociates to I( 2 P 3/2 ) + I( 2 P 1/2 ), which is 7589 cm 1 above...
-
The SO 2 molecule, analogous to H 2 O, has a 1 A 1 ground electronic state with the valence electronic configuration (1a 2 ) 2 (4b 2 ) 2 (6a 1 ) 2 (2b 1 ) 0 and low-lying excited singlet states of...
-
A spacecraft is traveling with a velocity of v0x = 5480 m/s along the +x direction. Two engines are turned on for a time of 842 s. One engine gives the spacecraft an acceleration in the +x direction...
-
Indicate whether the business activity noted below is an event or a transaction. Hired an advertising firm to develop a TV ad for a new product. The ad will be finished next month and you will pay...
-
Prepare for the inevitablethe day you do not know an answer. Sometimes, it feels embarrassing to ask questions at work. In the following situations, which questions are im-portant to ask at work, and...
-
The following statements are about investment companies and the Investment Company Act of 1940. Select the answer choice containing the correct statement. An insurance company that is primarily...
-
1.00 kilogram of liquid water at 50.0 C is converted completely to ice at -50.0 C . What is the change in entropy for the water. (Answer in kJ/K with correct sign)?
-
(1) Explain what is absolute advantage and what is comparative advantage? (2) Using the information given in the table to demonstrate the pattern of advantages between the two countries for the two...
-
Refer to the preceding exercise and use only the skull breadths from 4000 B.c. and 150 A.D. Use the Wilcoxon rank-sum test with a 0.05 significance level to test the claim that the two samples are...
-
Using Apple, demonstrate how the differentiation strategy can be well implemented.
-
Identify all products expected for each of the following reactions. Take stereochemistry into account, and draw expected stereoisomer(s), if any: (a) (b) (c) (d) NBS hv
-
Ambien TM is a sedative used in the treatment of insomnia. It was discovered in 1982 and brought to market in 1992 (it takes a long time for new drugs to undergo the extensive testing required to...
-
The following compound is known to be chiral. Draw its enantiomer, and explain the source of chirality. CH
-
Calculate the Target Corporation's Cash Conversion Cycle for fiscal year 2 0 2 1 ( ending January 2 9 , 2 0 2 2 ) . Note: Use the Accounts Receivable balance included in Other Assets ( as defined in...
-
The figure has permieter P=56. Find the value of x. The value of x is (Type a whole number.) +2 A x+6 (Figure is not to scale)
-
During the COVID-19 pandemic, medical sales professionals could reasonably state, "If you don't buy these N-95 masks today, there may not be any tomorrow." What type of close were they using?

Study smarter with the SolutionInn App


