Find the measure of the indicated exterior angle in each of the triangles in Problems 1722. X
Question:
Find the measure of the indicated exterior angle in each of the triangles in Problems 17–22.
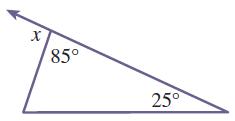
Transcribed Image Text:
X 85 25
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Muqadas Javed
I am a mentor by profession since seven years. I have been teaching on online forums and in universities. Teaching is my passion therefore i always try to find simple solution for complicated problems or task grasp them so that students can easily grasp them.I will provide you very detailed and self explanatory answers and that will help you to get good grade. I have two slogans: quality solution and on time delivery.
4.60+
24+ Reviews
144+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Find the measure of the indicated exterior angle in each of the triangles in Problems 1722. X 65 60 55
-
Find the measure of the indicated exterior angle in each of the triangles in Problems 1722. x 80 70 30
-
Find the measure of the indicated exterior angle in each of the triangles in Problems 1722. 100 20
-
Using the SEDAR database, find the most recent annual reports for two Canadian retailers (e.g., Loblaw, Rona, Danier Leather). Required: a. Based on the information provided in the companies audited...
-
Three wooden beams, each of length of 3a, are nailed together to form the support system shown. Assuming that only vertical forces are exerted at the connections, determine the vertical reactions at...
-
Compute the pseudo inverse of A. A = -1
-
FRAUD PREVENTION AND DETERRENCE IN ACTION Assume the following facts: thirty-day collection period. The invoice is used to post-sales to the accounting system. Checks are received in the mailroom and...
-
Ashkar Company ordered a machine on January 1, 2012, at an invoice price of $21,000. On the date of delivery, January 2, 2012, the company paid $6,000 on the machine, with the balance on credit at 10...
-
Calculate inventory amounts when costs are declining During the year, Hooker Incorporated has the following inventory transactions. Date January 1 Transaction Beginning inventory March 4 Purchase...
-
Using only a straightedge and a compass, reproduce the triangles given in Problems 2328.
-
Find the measure of the indicated exterior angle in each of the triangles in Problems 1722. 43 52 X
-
Effect of order quantity on special order decision Lang Company made 100,000 electric drills in batches of 1,000 units each during the prior accounting period. Normally, Lang markets its products...
-
In counting its year-end inventory, Oops Company double-counted the merchandise located in one of its warehouses. Will ending inventory be overstated (O) or understated (U) or OK as a result of this...
-
ARAMCO has a dividend growth rate of 0.0495% from year 2019-2022. The company paid a dividend of SAR 0.3518 per share for each quarter in year 2021. Assuming a required rate of return of 3.5%, answer...
-
Is there a relationship between a CEO's retirement and the R&D expenses in a firm? Explain.
-
Create, compile, and run a program that performs a simulation. Using the code in the book, simulate a grocery checkout checkout counter with one line. People will enter the line. The only thing a...
-
-> = A disk of mass m = 70.0 g and radius "1 = 3.50 cm slides on a frictionless sheet of ice with velocity v, where v = 11.00 m/s, as shown in a top-down view in figure (a) below. The edge of this...
-
Using the most current annual reports or the Forms 10-K for Pfizer, one of the worlds largest pharmaceutical manufacturers, and Walgreens, the drugstore chain, complete the requirements below. To...
-
At the beginning of its fiscal year, Lakeside Inc. leased office space to LTT Corporation under a seven-year operating lease agreement. The contract calls for quarterly rent payments of $25,000 each....
-
What physical quantities are governed by the quantum numbers of an atomic electron?
-
Why does the energy of an atomic electron depend on its angular momentum and magnetic quantum numbers as well as on its principal quantum number?
-
Can more than one electron occupy an atomic orbital? Is there a limit to how many?
-
Explain the different types of financial statements simply? Explain all types briefly
-
Coronado Inc. issued $ 9 2 0 , 0 0 0 of 1 0 % , 1 0 - year bonds on June 3 0 , 2 0 2 5 , for $ 8 1 4 , 4 7 2 . This price provided a yield of 1 2 % on the bonds. Interest is payable semiannually on...
-
Actual pounds (g) Revenue ($4.25g) Quilcene Oysteria Flexible Budget For the Month Ended August 31 Expenses: Packing supplies ($0.35g) Oyster bed maintenance ($3,300) Wages and salaries ($2,400 +...

Study smarter with the SolutionInn App


