Twelve people serve on a board and are considering three alternatives: A, B, and C. Here are
Question:
Twelve people serve on a board and are considering three alternatives: A, B, and C. Here are the choices followed by vote:
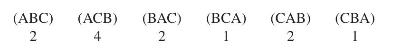
Determine the winner, if any, using the voting methods in Problems 39-44.
Majority rule none
Transcribed Image Text:
(ABC) (ACB) 2 4 (BAC) 2 (BCA) 1 (CAB) 2 (CBA) I
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Arshad Ahmad
Well, I am really new to tutoring but I truly believe a good student can be a better teacher. I have always been a topper at school. I passed my Chartered Accountancy at a very young age of 23, a rare feat for most of the students. I am really dedicated to whatever work I do and I am very strict regarding deadlines. i am always committed and dedicated to whatever work allotted to me and I make sure it is completed well within deadline and also I try to give my best in whatever I do. Hope we will have a good time studying together.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Twelve people serve on a board and are considering three alternatives: A, B, and C. Here are the choices followed by vote: Determine the winner, if any, using the voting methods in Problems 39-44....
-
Twelve people serve on a board and are considering three alternatives: A, B, and C. Here are the choices followed by vote: Determine the winner, if any, using the voting methods in Problems 39-44....
-
Twelve people serve on a board and are considering three alternatives: A, B, and C. Here are the choices followed by vote: Determine the winner, if any, using the voting methods in Problems 39-44....
-
Robert is the owner of an automobile manufacturing company. He calls for a board meeting and tells his directors that he wants to build a car that lets the users experience power and exhilaration. He...
-
Liquid nitrogen with a mass of 100 g at 77.3 K is stirred into a beaker containing 200 g of 5.00C water. If the nitrogen leaves the solution as soon as it turns to gas, how much water freezes? (The...
-
D. Tomlinson Retail seeks your assistance in developing cash and other budget information for May, June, and July. The store expects to have the following balances at the end of April: Cash...
-
Refer to the extracts from annual report 2005-06 of the Whirlpool of India Limited regarding its fixed assets, related significant accounting policies and profit/loss on disposal thereof as...
-
You are the manager of a used car firm known as Reliant Motor Company. Your lawyer has called to tell you that John Doe, a customer you have been dealing with for several months, has filed a lawsuit...
-
Eve Co purchases a machine that had a list price of $10,000 on January 1, 2020 The machine has no estimated residual value and a 2 years estimated useful life. The Company is using the...
-
In the apportionment of the House of Representatives based on the 1790 census , there are 15 states. At that time, Maine was still considered part of Massachusetts. If Maine had been a separate...
-
In 1993 the 101st International Olympic Committee met in Monaco to select the 2000 Winter Olympics site. The cities in the running were Beijing (B), Berlin (L), Istanbul (I), Manchester (M), and...
-
Write explicitly the \(2 \times 2\) matrices \(g=e^{i \alpha_{i} \sigma_{i} / 2}\), where \(\sigma_{i}\) are the Pauli matrices, and compare with \(g(\theta, \phi, \psi)\) for \(S O(3)\), to find an...
-
Calculate the price elasticity for a $1 change in price at initial price level $300 for the demand function Qd = 1,500 1.5 P .
-
From the following figures calculate the closing inventory-in-trade that would be shown using (i) FIFO, (ii) LIFO, (iii) AVCO methods. March September Bought 200 at 20 each 100 at 22 each December...
-
F. Brown drew up the following trial balance as at 30 September 2011. You are to draft the income statement for the year ending 30 September 2011 and a statement of financial position as at that...
-
The credit sales and purchases for the month of December 2007 in respect of G. Bain & Co were: 2013 Dec 1 Sales to H. Impey Ltd 4 Sales to B. Volts 5 Purchases from G. Sharpe and Co 8 Purchases from...
-
For Question 29.1 draw up the trading account part of the income statement for the year showing the gross profits that would have been reported using (i) FIFO, (ii) LIFO, (iii) AVCO methods. Question...
-
Suppose that, as an owner of a federally insured S&L in the 1980s, the price of real estate falls, and most of your loans go into default. In fact, so many loans go into default that the net worth of...
-
Suppose the concentration of glucose inside a cell is 0.1 mm and the cell is suspended in a glucose solution of 0.01 mm. a. What would be the free energy change involved in transporting 10-o mole of...
-
A two-stage compressor having an intercooler takes in air at 300 K, 100 kPa, and compresses it to 2MPa, as shown in Fig. P7.45. The cooler then cools the air to 340 K, after which it enters the...
-
A flow of 5 kg/s water at 100 kPa, 20C should be delivered as steam at 1000 kPa, 350C to some application. We have a heat source at constant 500C. If the process should be reversible, how much heat...
-
An adiabatic air turbine receives 1 kg/s air at 1500 K, 1.6 MPa and 2 kg/s air at 400 kPa, T 2 in a setup similar to that with an exit flowat 100 kPa. What should the temperature T 2 be so that the...
-
7. (12 Points) State what takes place when each of the following instructions reach the top of ROB and their operands are all available:+ a) fld b) fadd wwwww f2, 0(x6)+ f2, f4, f6+ c) fsd f2, 0(x2)+...
-
Following are two examples of C code. Translate the C code to MIPS assembly code. Use a minimum number of instructions. Assume that the value a, b, c, d, i are in registers $s0, $s1, $s2, $s3, $t0...
-
Analyze the truss below using "Method of Joints" and determine internal axial forces in each member of the truss. PINNED SUPPORT 115k lok lok lok VE F 10' 10' * 10' 30' 01 115k ROLLER SUPPORT

Study smarter with the SolutionInn App


