Question: Using the relation 1/v g = dκ/dv, prove that v dv v? dv Vg
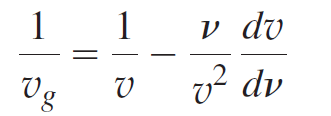
v dv v? dv Vg
Step by Step Solution
3.45 Rating (165 Votes )
There are 3 Steps involved in it
1u g d... View full answer

Get step-by-step solutions from verified subject matter experts


