Compound X (C 5 H 10 O) shows a strong IR absorption band near 1710 cm 1
Question:
Compound X (C5H10O) shows a strong IR absorption band near 1710 cm−1. The broadband proton-decoupled 13C NMR spectrum of X is shown in Fig. 2. Propose a structure for X.
Fig. 2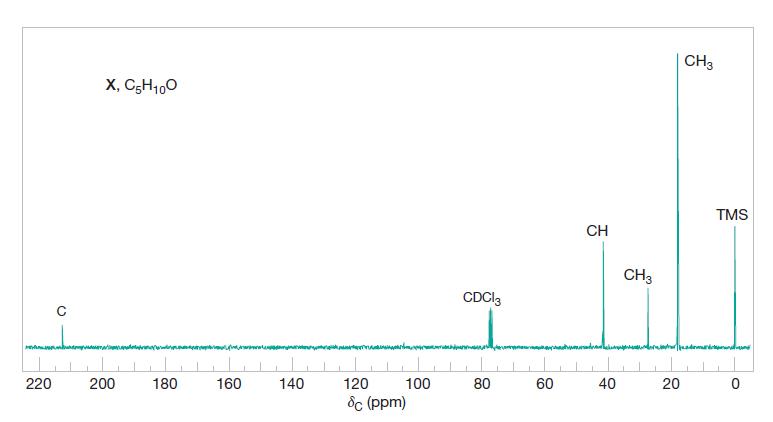
Transcribed Image Text:
220 C X, C5H100 200 180 160 140 100 120 8c (ppm) CDC13 80 60 CH 40 CH3 20 CH3 TMS dans 0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
Based on the IR absorption band at 1710 cm1 we can infer that compound X contains a carbonyl functio...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Compound T (C5H8O) has a strong IR absorption band at 1745 cm-1. The broad-band proton decoupled 13C spectrum of T shows three signals: at d 220 (C), 23 (CH2), and 38 (CH2). Propose a structure for T.
-
The 1H NMR spectrum of 1-chloropentane is shown at 60 MHz (spectrum H) and 500 MHz (spectrum I), Explain the differences in appearance of the two spectra, and assign the signals to specific hydrogens...
-
The proton decoupled 13 C NMR spectrum of pyridine will display how many peaks? (a) One; (b) Two; (c) Three; (d) Four; (e) Five.
-
Assume a retailer has fixed costs of $10,000, a unitvariable cost of $25, and a 50% retail margin. How many units must be sold for her tobreak-even? If she has a target profit of $200,000, how many...
-
5/3 1/6 x < 1/2 Describe the solution set as an inequality, in interval notation, and on a graph.
-
Such factors as regulations, competition, and corporate actions relate to: A. fund-closure risk. B. counterparty risk. C. expectation-related risk.
-
At a political debate, a sample of audience members were asked to name the political party to which they belonged. Their responses are shown in the table. What is the mode of the responses? Political...
-
The beta coefficient for Stock C is bC = 0.4 and that for Stock D is bD = 0.5. (Stock Ds beta is negative, indicating that its rate of return rises whenever returns on most other stocks fall. There...
-
What are mashups? How do they enable social media applications? 5-4. How can social software help harness the wisdom of the crowd? 5-6. How can social software enhance communication? 5-8. What is a...
-
Using the alternative-parameter method, determine the parameters of the following distributions based on the given assessments. Refer to Step 5.5 if necessary. a. Find the parameter value for the...
-
There are nine stereoisomers of 1,2,3,4,5,6-hexachlorocyclohexane. Seven of these isomers are meso compounds and two are a pair of enantiomers. (a) Write structures for all of these stereoisomers,...
-
For each of the following questions, please provide a route that could reasonably be expected to convert the starting material into the final product. In each case, more than one reaction is...
-
Baker Company Limited (BCL) was founded in 2023. Its first year of operations turned out to be a good one, as far as start-up years go. The company not only broke even but actually showed a very...
-
Complete the model by filling in the blank cells before answering the question below. Answers should be rounded to the nearest whole number, comma separating 000s, NOT written in currency format. So...
-
You would like to borrow $25,000 from a local bank.You will make equal monthly payments over a fifteen-year period.What will your monthly payment be if yearly interest rate is 6%? A company is...
-
Simon has current wealth of $36, including $20 in cash. With probabil- ity Simon's money will be stolen (contingency 1), leaving him with only $16 to spend on consumption, and with probability his...
-
Last year Neepawa Industries Ltd. reported earnings per share of $1.35 and paid a dividend equal to one-third of its earning per share. If earnings and dividends are expected to grow at a constant...
-
Vira was sole shareholder of Koltana, Inc., which held a $1,000,000 term insurance policy on Vira's life payable to her son Ivan. Two years ago, when the stock of Koltana was worth $4,000,000, Vira...
-
Suppose that S = [v1, v2, v3} is a linearly dependent set of vectors in a vector space V. Is T = {w1, w2, w3}, where w1 = v1, w2 = v1, + v3, w3 = v1, + v2 + v3, linearly dependent or linearly...
-
3.16. For a system with non-identical service rates (see Sect. 3.5) and a limit of N jobs in the system (Eq. 3.13), obtain an expression for the mean service time per job, E[Ts], as a function of the...
-
Indicate whether each of these objects is chiral or achiral: (a) Golf ball (b) Baseball glove (c) Clock (d) T-shirt (e) Dress shirt (f) Automobile
-
Determine whether each of these molecules is chiral, for those that are chiral, put an asterisk at the chirality center. b) a) d) I e) )
-
Indicate whether each of these objects or molecules has a plane of symmetry: c) Ear b) Pencil a) Idealized human face I CH3 e) f) d) CH3 . Cl Br CH3 "H. CH3 "H g) h) Cl
-
A scuba diver is exploring a shipwreck at a depth of 30 meters in the ocean. The diver's tank contains 5.0 moles of compressed air at a temperature of 25C. What is the pressure of the air in the tank...
-
1. Determine the atmospheric pressure at a location where the barometric reading is 720 mm Hg. 2. A pressure gauge connected to a tank reads 52 psi at a location where the barometric reading is 29.6...
-
Assume that a bond has an annual coupon rate of 6.80 percent, but makes coupon payments on a semi-annual basis, has a par value of $1,000, a yield to maturity of 7.20 percent, and a current price of...

Study smarter with the SolutionInn App


