For each of the following complexes, determine the oxidation state of the metal, the total number of
Question:
For each of the following complexes, determine the oxidation state of the metal, the total number of valence electrons it possesses, and state whether the metal is coordinatively saturated or unsaturated.
(a)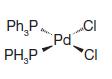
(b)
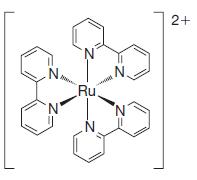
(c)
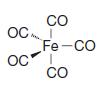
Transcribed Image Text:
Ph₂P PH₂P Pd CI CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 84% (13 reviews)
a The palladium atom in the elemental state has 10 valence electrons The charge on the complex as a ...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Alisha has a February starting balance of $678.98 in her checking account. During the month, she made deposits that totaled d dollars and wrote checks that totaled c dollars. Let E = her ending...
-
For each of the following complexes, determine the oxidation state of the transition-metal atom. a. [CoCl(en)2(NO2)]NO2 b. [PtCl4]2 c. K3[Cr(CN)6] d. [Fe(H2O)5(OH)]2+
-
State the research problems for each of the following management decision problems. a. Should a new product be introduced? b. Should an advertising campaign that has run for three years be changed?...
-
The standard manufacturing process for thin-film resistors produces resistors with an average of 1k and a standard deviation of 3.5 Q. Twenty samples of a new process were tested, and the data can be...
-
The pedestrian and bicyclist fatality rates in 10 randomly selected large cities are shown in Table 68. Let p be a citys pedestrian fatality rate (in number of deaths per 10,000 walking commuters),...
-
An instanton is a quantum mechanical excitation that is localized in space like a particle. They are closely related to solitary waves or solitons that were first observed in the mid-nineteenth...
-
An analysis of the accounts of Small Appliances Pty Ltd reveals the following manufacturing cost data for the month ended 30 June 2025. Required (a) Prepare the cost of goods manufactured schedule...
-
Hart Company sells and delivers office furniture across Western Canada. The costs associated with the acquisition and annual operation of a delivery truck are given below: Insurance. . . . . . . . ....
-
HISTORY OF PSYCHOLOGY PART 1 : Phrenology was a wildly popular but untrue scientific theory. Find another popular but untrue scientific theory. Find a URL on the internet exploring at least one more...
-
Match the following and select the correct option. Height of Binary search tree(in worst o([log ]-1) 1 a (2n+1) case) Height of Ternary tree b O(n) Height of B-tree(when c minimum degree, -2) (log,...
-
The sedative-hypnotic ethinamate can be synthesized by the following route. Provide structures for ethinamate and the intermediates A and B: Cyclohexanone (1) HC=CNa, (2) HO+ A (CH12O) CICOCI B...
-
Heating acetone with an excess of phenol in the presence of hydrogen chloride is the basis for an industrial process used in the manufacture of a compound called bisphenol A. (Bisphenol A, about...
-
Yussuff Company manufactures CH-21 through two processes: mixing and packaging. In July, the following costs were incurred. ____________________________________Mixing _______ Packaging Raw materials...
-
Fourier analysis tells us that a square wave y(t) with period and amplitude of one is given by the following sum of sine waves: y(t) = TU n sin (2nt) n n = 1,3,5,7,...,00 Write a VI that approximates...
-
The July 31 bank statement for a catering company reported a balance of $15,014. The company's book balance shows $19,960. Two checks written by the company are outstanding, #333 and #336 in the...
-
Suggest a suitable design for a pair of helical gears to transmit 100 hp between a 2400 rpm electric motor and an 800-rpm load that is essentially free of shock. Forty hour-per-week operation is...
-
Consider a homogeneous product Cournot oligopoly consisting of n ex-ante symmetric firms who face the linear inverse demand: p=a- 9i IEN where p is the price, q; is firm i's output and a > 0. The...
-
If estimate overhead is $500, total allocated overhead for the period was $525 and actual overhead for the period was $510, is overhead over or underapplied and by how much?
-
Simplify the following. (a) 4(3u - v + w) - 2[(3u - 2v) - 3(v - w)] + 6(w - u - v)
-
In the current year, the City of Omaha donates land worth $500,000 to Ace Corporation to induce it to locate in Omaha and create an estimated 2,000 jobs for its citizens. a. How much income, if any,...
-
Provide names for thesecompounds: SH SH b) H OS e) CH3 SOCH,CH,CH, d)
-
Draw the structures for these compounds: (a) 2-Butanethiol (b) Benzenethiol (c) Isopropyl methanesulfonate (d) p-Bromobenzenesulfonic acid (e) Phenyl trichloromethyl sulfide
-
Provide systematic names for these compounds: NH2 ) H,, b) a) CO,H Leucine (an amino acid) CH OCH3 d) H. NHCH, CN h) g) CH,CCH,C=N Ph
-
You obtained job-related information about the HR assistant position from O*Net. Answer the following sections based on that information. QUESTIONS: The following is the list of questions included in...
-
Marcia Stanton is the new manager of the materials storeroom for Taylor Manufacturing. Marcia has been asked to estimate future monthly purchase costs for part #696, used in two of Taylor's products....
-
Miller Company's contribution format income statement for the most recent month is shown below: Sales (37,000 units) Variable expenses Contribution margin Fixed expenses Net operating income...

Study smarter with the SolutionInn App


