Predict the outcome of the following reactions: CN CI 2 equiv. KNH lig. NH3, 33 C
Question:
Predict the outcome of the following reactions:
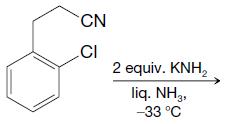
Transcribed Image Text:
CN CI 2 equiv. KNH₂ lig. NH3, 33 C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
For the first reaction the expected outcome is the formation of 1hexanol This is because the reactan...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Predict the outcome of the reactions represented by the following equations by using the activity series, and balance the equations. (a) Cu(s) + HCl(aq) (b) 12(s) + NaBr(aq)- (c) Mg(s) + CuSO (aq...
-
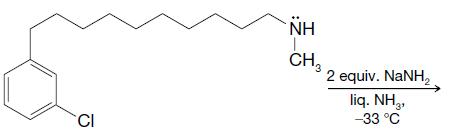
Predict the outcome of the following reactions: (a) (b) CN CI 2 equiv. KNH2 liq. NH3 -33 C NH CH3 2 equiv. NaNH liq. NH2 -33C Cl
-
Predict the outcome of the following reaction. -NH2
-
The Kc for the following reaction is 9.30 X 10^-2 at 25C:PCl5(g) <-> PCl3(g) + Cl2(g) How many moles & grams of PCl5 must be added to a 2-literflask to obtain a Cl2 concentration of 0.150M...
-
X 3 1 Describe the solution set as an inequality, in interval notation, and on a graph.
-
A man is found shot to death in the front seat of his car. All the windows are closed and the doors are locked; there are no bullet holes anywhere in the car and he did not commit suicide. How was he...
-
Draw a frequency polygon for the frequency distribution in Example 2. Describe any patterns. Data from Example 2 Using the frequency distribution constructed in Example 1, find the midpoint, relative...
-
Munoz Corporation incurs the following annual fixed costs: Item Cost Depreciation .......... $ 75,000 Officers salaries ........ 160,000 Long-term lease ....... 38,000 Property taxes ......... 12,000...
-
In the context of absorptive separation processes, how does the introduction of reactive absorption (e.g., using chemical reactants) alter the absorption equilibrium and kinetics? What are some...
-
Four different proprietorships, Jupiter, Mars, Saturn, and Venus, show the same balance sheet data at the beginning and end of a year. These data, exclusive of the amount of owner's equity, are...
-
Provide a mechanism for the following reaction. NO NO 2 ss-sr NaOCH3 OH NO O=5 S=0 NO
-
Starting with aniline, outline a synthesis of each of the following: (a) p-Bromoaniline (b) o-Bromoaniline (c) 2-Bromo-4-nitroaniline (d) 4-Bromo-2-nitroaniline
-
Should other employees (cleaner/spotters, counter people) be put on a similar plan? Why or why not? If so, how exactly?
-
168. is responsible for graft rejection : (1) Antibody mediated immunity (2) Cell mediated immunity (3) Innate immunity (4) Herd immunity
-
QUESTION TWO a) State and explain five monetary policy instruments used by the central bank to regulate interest rate, inflation rate, exchange rate and the quantity of money in the economy [15...
-
Simpson, age 4 5 , is a single individual who is employed full time by Duff Corporation. This year Simpson reports AGI of $ 5 5 , 2 0 0 and has incurred the following medical expenses: Dentist...
-
Bonds payable record issuance and premium amortization Jessie Co . issued $ 3 million face amount of 9 % , 2 0 - year bonds on July 1 , 2 0 2 2 . The bonds pay interest on an annual basis on June 3 0...
-
Currently, Stock A pays a dividend of $2.00 a share, and you expect the dividend to grow rapidly for the next 4 years at 20%. After this initial period of super growth, the rate of increase in the...
-
(a) Show that C[a, b] is a real vector space. (b) Let W (k) be the set of all functions in C[a, b] with ((a) = k. For what values of k will W (k) be a sub-space of C[a, b]? (c) Let t1, t2, ( ( ( ( tn...
-
The Taylor's series expansion for cosx about x = 0 is given by: where x is in radians. Write a user-defined function that determines cosx using Taylor's series expansion. For function name and...
-
How might the structure of DDT be modified to make it again effective against resistant insects?
-
Explain which product is formed when each of these alkyl chlorides reacts with sodium ethoxide in ethanol.
-
Explain which of these alkyl chlorides reacts faster with sodium ethoxide in ethanol.
-
Whatare basis of the segmentation? How would you improve on this strategy in marketing ? What are the basis of the segmentation? How would you improve on this strategy?
-
Jimmy has a bond with a $1,000 face value and a coupon rate of 9.5% paid semiannually. It has a five-year life. a) If investors are willing to accept a 14 percent rate of return on bonds of similar...
-
A zero-coupon bond that will pay $1,050 in 12 years is selling today for $441.91. What interest rate does the bond offer?

Study smarter with the SolutionInn App


