Predict the products from each of the following reactions. (a) (b) (c) (d) (e) (f) (g) (1)
Question:
Predict the products from each of the following reactions.
(a)
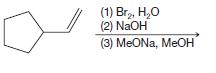
(b)
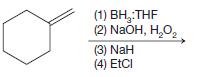
(c)
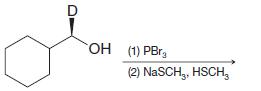
(d)
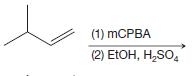
(e)
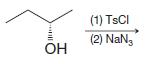
(f)
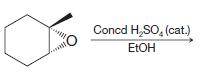
(g)
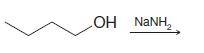
Transcribed Image Text:
(1) Br₂, H₂O (2) NaOH (3) MeONa, MeOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
A sample of copper sulphate pentahydrate contains 3.782 g of Cu. How many grams of oxygen are in the sample? 1) 0.952 g 1 2) 3.809 g 3) 4.761 g 4) 8.576 g
-
Predict the products from each of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) Hl (excess) Hi H,so,,H20 O MeONa O MeOH, cat. H2SO (1) EtSNa (2) H2o HCl (1 equiv.) MeONa (1)EtON...
-
1. [10] Let A = 2 4 a b c d e f g h i 3 5, B = 2 4 d + 5g e+ 5h f+ 5i a 2g b 2h c 2i g h i 3 5, C = 2 4 2g 2h 2i 3d 3e 3f 1a 1b 1c 3 5. Suppose that det A = 5. Find det B = , det C = , and det (AC) =
-
Two moles of an ideal monatomic gas go through the cycle abc. For the complete cycle, 800 J of heat flows out of the gas. Process ab is at constant pressure, and process bc is at constant volume....
-
How are communicable diseases devastating human and economic development in some poor nations?
-
After stocktaking for the year ended 31 May 2016 had taken place, the closing inventory of Cobden Ltd was aggregated to a figure of 87,612. During the course of the audit which followed, the...
-
For symmetrical blading, the degree of reaction is _____.
-
Selected financial information of Gray Plains Inc. for the current year follows. During the year, Gray Plains Inc. took advantage of the current interest rates and called $125 million of long-term...
-
Olive You applies manufacturing overhead at a rate of $0.40 per kilowatt hour (kWh) of electricity. Olive You's production facility used 125,000 kWh of electricity during January. The following...
-
BigCos chief financial officer is trying to determine a fair value for PrivCo, a nonpublicly traded firm that BigCo is considering acquiring. Several of PrivCos competitors, Ion International and...
-
Draw a free-energy diagram (reaction progress versus free energy) for each of the following reactions. Be sure to label the axes, transition state(s), the energy of activation for each step, and H,...
-
Verizon Communications, Inc. is one of the worlds largest providers of communication services. The following information, taken from the companys annual reports, is available for the years 2018,...
-
Murray, Inc., purchased a new inventory item two times during the month of April, as follows: Apr. 5 100 units @ $5.00 Apr. 15 100 units @ $5.05 a. What is the amount of the ending inventory of this...
-
Assume the salary scale given in Table 10.1 of AMLCR 2nd edition and a valuation date of 1 January. (a) A plan member aged 40 at valuation received $80,000 in salary in the year to the valuation...
-
While high dividends are preferred by investors, this is not necessarily a good thing. Explain the situation what is the high dividends should not be considered?
-
Price Level AS Long-run aggregate supply Short-run aggregate supply AS Aggregate demand Long Run Equilibrium The question starts with the economy in Long Run Equilibrium at the Natural Rate of...
-
Using the FCF method, what is the most likely NPV of this project? Would you accept it?
-
Describe the two unit of payment strategies that UK uses to contain costs compared to the US.You must explain how this affects the components of the iron triangle in UK?
-
In problems 1-3 find the equation of the circle satisfying the given conditions. 1. Center (1, 1) radius 1 2. Center (-2, 3), radius 4 3. Center (2, - 1), goes through (5, 3)
-
As you rewrite these sentences, replace the cliches and buzzwords with plain language (if you don't recognize any of these terms, you can find definitions online): a. Being a jack-of-all-trades, Dave...
-
While working in the chemical stockroom, you discover an unlabeled bottle containing a liquid compound. You carefully smell the liquid and discover that it has a fishy odor. What functional group do...
-
Turpentine, obtained from pine trees, is composed primarily of ?-pinene and ?-pinene, Explain whether you expect turpentine to mix with water. If a point dissolves in turpentine, what does this...
-
The structure of a typical fat is shown here. Estimate the energy content of fat compared to the other compounds discussed in the Focus On box on p. 146 and explain yourreasoning. CH OC(CH) 16CH3 O...
-
1- The binomial distribution N! B(M) = (N-M)!M! describes the probability of placing M identical particles into N equivalent physical sites, with a limit of one particle/site if the probability of a...
-
Margo, a calendar year taxpayer, paid $1,580,000 for new machinery (seven-year recovery property) placed in service on August 1, 2023. Required: a. Assuming that the machinery was the only tangible...
-
CASA (Australia's Civil Aviation Safety Authority) and I CAO SMS (Safety Management Systems) and the alphanumeric risk categorization system for an aviation risk register. *** What I need help with...

Study smarter with the SolutionInn App


