Question: Using the (E )(Z ) designation [and in parts (e) and (f) the (R )(S ) designation as well give IUPAC names for each of
Using the (E )–(Z ) designation [and in parts (e) and (f) the (R )–(S ) designation as well give IUPAC names for each of the following:
(a)
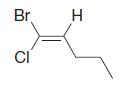
(b)
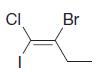
(c)
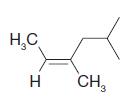
(d)
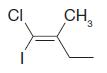
(e)
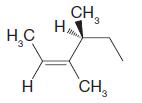
(f)
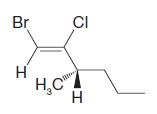
Br H CI
Step by Step Solution
3.60 Rating (178 Votes )
There are 3 Steps involved in it
If the two groups with the higher priorities are on the same ... View full answer

Get step-by-step solutions from verified subject matter experts


