What starting materials could be used to synthesize the following compound by a SuzukiMiyaura coupling? H.
Question:
What starting materials could be used to synthesize the following compound by a Suzuki–Miyaura coupling?
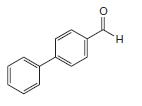
Transcribed Image Text:
H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
Solution The starting materials for this method are an olefin a boronic acid and copper II c...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
What starting materials could be used to synthesize each of the following compounds by a Sonogashira coupling reaction? (a) (b) CI
-
Synthesize the following compound starting with ethyne and 1-bromopentane as your only organic reagents (except for solvents) and using any needed inorganic compounds. Br Br
-
Synthesize the following compound by a method that derives all carbon atoms from alcohols of five carbons or fewer. You may use any other reagents needed. OH
-
A copper-zinc alloy has the following properties: Grain diameter (mm) Strength (MPa) 0.015 .................................. 170 MPa 0.025 .................................. 158 MPa 0.035...
-
The windchill (or windchill factor) is a measure of how cold you feel as a result of being exposed to wind. Table 64 pro- vides some data on windchills for various temperatures when the wind speed is...
-
Students of the author collected a simple random sample of times (sec) of wristwatch errors, and a few of those times are listed below. Negative values correspond to watches that are running ahead of...
-
Smart Manufacturing Systems Pty Ltds accountant recently prepared the following data from the companys accounting records for the year ended 30 June 2025. Required (a) Prepare a cost of goods...
-
The following facts apply to Walken Company during December 2011: a. Walken began December with an accounts receivable balance (net of bad debts) of 25,000. b. Walken had credit sales of 85,000. c....
-
Galileo dropped two balls from the Leaning Tower of Pisa. The tower is about 56 m tall. For the questions below, assume the mass of one of the balls is 10 kg and the mass of the second ball is 18 kg....
-
The position of a particle as a function of time is given by r(vector) = (5.0i + 4.0j)t 2 m, where t is in seconds. a. What is the particles distance from the origin at t = 0, 2, and 5 s? b. Find an...
-
What is the product of each of the following reactions? (a) (b) (c) (d) SnBu3 Pd catalyst
-
Starting with 1-butanol and using any other required reagents, outline a synthesis of each of the following compounds. You need not repeat steps carried out in earlier parts of this problem. (a)...
-
Find the center of mass and the moment of inertia about the y-axis of a thin rectangular plate cut from the first quadrant by the lines x = 6 and y = 1 if (x, y) = x + y + 1.
-
What you need to do: Write and test the method public static String capitalize (String str) which takes a string argument and returns a new String consisting of str, with the first character in...
-
(1) Tesla had stated that its annual production target for 2030 is 30 million electric vehicles. (a) What are the key constraints that Tesla faces in trying to achieve this target? (b) Is this...
-
Has the price of any of the products or services your company produced changed recently? How could you use elasticity of demand to help in making that decision? Price elasticity of demand". You can...
-
Ronald owns a row-boat store. In his accounting records, he included his personal computer and all of his personal rowing gear. Ronald is violating what principle of accounting? Explain briefly with...
-
Accounting Inc. has ordered imports from Uruguay, and its imports are invoiced in Uruguayan pesos. The dollar value of the payables (based on today's exchange rate) from its imports during this year...
-
Let v, v1,..., vn denote vectors in a vector space V and let a, a1,..., an denote numbers. Use induction on n to prove each of the following. (a) a(a1 + a2 + ... + an)v = a1v + a2v + ... + anv
-
Write the statement to store the contents of the txtAge control in an Integer variable named intAge.
-
Explain which compound has the higher melting point.
-
Explain which compound has the higher boiling point. Discuss in detail.
-
Explain whether each compound is soluble in aqueous NaOH, aqueous NaHCO3 both, or neither.
-
es Hart, Attorney at Law, experienced the following transactions in Year 1, the first year of operations: 1. Accepted $16,600 on April 1, Year 1, as a retainer for services to be performed evenly...
-
Dahlia Corporation has a current accounts receivable balance of $439,516. Credit sales for the year just ended were $5,503,810. a. What is the receivables turnover? Note: Do not round intermediate...
-
Why does the organizational structure hold political significance? Provide an in-depth analysis of this concept using examples from both academic literature and real-world instances. Additionally,...

Study smarter with the SolutionInn App


