Working backwards, deduce the starting material that led to the indicated product through the defined reactions. (a)
Question:
Working backwards, deduce the starting material that led to the indicated product through the defined reactions.
(a)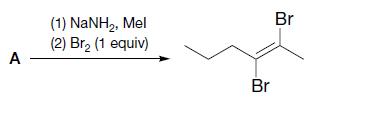
(b)
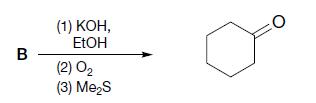
(c)
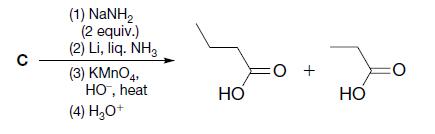
Transcribed Image Text:
Br (1) NaNH,, Mel (2) Br, (1 equiv) A - Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)

Answered By

Djordje Surdanovic
Chemistry seems like a boring, difficult, and arduous science, like other natural sciences. However, what if I told you that each of us has actually been a scientist since we were little? You must have wondered why the sun exists, why the water is liquid, the grass is green and the sky is blue? So did I, but never got the right answer explained in a suitable and interesting way. Therefore, I asked myself why couldn't I become an educator and try to improve and modify the long-existing school method. Just as my teen years began I took interest in guitar playing and was always fascinated by the tones. The vibrating of the strings made me wonder, what causes the sound? I then researched and explored that certain vibrations caused by the movement of atoms are responsible. Well, then I got only deeper and deeper into it, and eventually fell in love with chemistry. In my early university days, I started teaching at some private primary schools, and my method turned out pretty well. I then upgraded and moved on to give private lessons to my colleagues. A teaching assistant noticed me and offered me a job as a freelancer, or in other words an online tutor. And so did my freelance career started. I then applied for several companies and got my first job. Since then I have been helping students all around the globe! So are you afraid of chemistry? Well, let's fight that fear and finally understand chemistry as a part of our everyday life, not just as a science. Everyone is welcome!
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Working backward, deduce the starting material that led to the indicated product through the defined reactions (A) (B) (1) NANH, (2 equiv) A (2) Li/NH,
-
Draw the structure of the starting material that would undergo azir-elimination give the -E isomer of the alkene product in the E2 reaction of Eq. 9.40.
-
The starting material for a commercial synthesis of vitamin C is l-sorbose (see the following reaction); it can be synthesized from d-glucose through the following reaction sequence: The second step...
-
Use (4.1), for a perfectly conducting fluid, and the nonlinear equation of continuity (1.1), to show that the change of B with time in a fluid element is related to changes of density according to...
-
Explain how each of the following is expected to affect growth: a. Increase in technology. b. Positive externalities. c. Patents. d. Learning by doing. e. Technological lock-
-
Archer Uniforms, Inc., is a distributor of professional uniforms to retail stores that sell work clothing to professionals, such as doctors, nurses, security guards, etc. Traditionally, most of the...
-
Describe the privileging and credentialing process.
-
On January 1, 2009, Pope Company purchased 90% of Sun Companys common stock for $5,800,000 cash. Immediately after the acquisition, the two companies balance sheets were as follows: Sun Companys note...
-
Dr. Wong is an equity portfolio manager with two new clients: The Hong Kong University Endowment Fund and the Tung Wah Group Foundation. The Hong Kong University Endowment Fund (the Fund) is overseen...
-
Complete Keith's tax return including all required schedules and forms using prince edward island as province, using the fillable forms package. Taxpayers Information Taxpayer #1 Name: Keith Dox...
-
In one industrial synthesis of ethanol, ethene first undergoes an addition reaction with sulfuric acid, and this product undergoes hydrolysis to ethanol. Write a mechanism for the addition of...
-
Given the following information, elucidate the structures of compounds A and B. Both compounds are soluble in dilute aqueous HCl, and both have the same molecular formula. The mass spectra of A and B...
-
At December 31, 2018, Percheron Inc. had a deferred tax asset of $30,000. At December 31, 2019, the deferred tax asset is $59,000. The company's 2019 current tax expense is $61,000. What amount...
-
Amy Dixon opened the Dixon Travel Agency in January, and the company entered into the following transactions during January: a On 2 January Amy deposited \(\$ 23000\) in the cash account. b To...
-
If a problem comes to light during the budgeting process, what is the manager likely to do?
-
Since the future is uncertain and circumstances are likely to change, why should the managers and owners of a business bother to plan?
-
If you just finished budgeting sales for next year, what information would you need to be able to budget cash collections from sales?
-
Your friend Maxine plans to supplement her job salary by running her own company at night and on the weekends. When the company earns enough money so that she can pay for a holiday house in Fiji, she...
-
Rank the following compounds in order of decreasing ease of removing a proton from a methyl group: CH3 CH3 CH3 CH2CH3
-
Rosalie owns 50% of the outstanding stock of Salmon Corporation. In a qualifying stock redemption, Salmon distributes $80,000 to Rosalie in exchange for one-half of her shares, which have a basis of...
-
Using the information available in Figure 4.2, predict the position of the equilibrium in these reactions; that is, predict whether there is a higher concentration of reactants or products present at...
-
Use the information in Figure 4.2 to predict the positions of the equilibria in the reactions in problem 4.4.
-
Draw diagrams like that in Figure 4.3 for the reactions in problem 4.9.
-
What choice is a multiple of 5? a) 93 b) 63 c) 65 d) 84
-
Determine how many tablets will be needed to give the dosage. Prepare a dosage of 6.4 mg using tablets with a strength of 1.6 mg.
-
(a). Company Inc., private consumer regulator firm, claims that the standard deviation of the rating of Formula One races in the town main car racing circuit was at least 15.34. (b). An outside...

Study smarter with the SolutionInn App


