Write the mechanism for the following Heck reaction that can explain the final alkene stereochemistry and why
Question:
Write the mechanism for the following Heck reaction that can explain the final alkene stereochemistry and why the Pd species can promote the reaction to go to completion in only catalytic quantities.
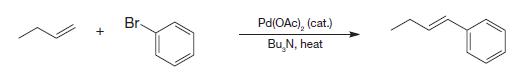
Transcribed Image Text:
+ Br- Pd(OAc), (cat.) Bu N, heat
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
Solution The mechanism for the Heck reaction is as follows Proposed final alkene stereochemistr...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Write the mechanism for the reaction of a cysteine residue with iodoacetic acid.
-
Write the mechanism for the formation of carbon tetrachloride, CCI4, from the reaction of methane with CI2 + hv.
-
Write the mechanism for the base-catalyzed formation of a cyclic ester from a 1,7-diester.
-
Jilmar Company acquired a delivery truck and made payment of P2,870,000 analyzed as follows: Price of truck - P2,500,000 Charge for extra equipment - P150,000 Value added tax (recoverable) -...
-
The mean amounts of savings various age groups believe are enough to have at retirement are shown in Table 78 Let s be the mean amounts of savings (in millions of dollars) a person at age a years...
-
Confidence level is 99%, is known to be $4,385,000, and the dotplot of 40 sample values of professional baseball players is as shown in Exercise 7. Assume that we want to construct a confidence...
-
For case 2 in Exercise 1, which country would prefer an international terms of trade of 1.2 rather than an international price of 1.66? Explain. Data from in exercise 1 For each of the following...
-
Use Worksheet 6.1. Rebecca Collins is evaluating her debt safety ratio. Her monthly take-home pay is $3,320. Each month, she pays $380 for an auto loan, $120 on a personal line of credit, $60 on a...
-
A 7400 kg rocket blasts off vertically from the launch pad with a constant upward acceleration of 2.20 m/s2 and feels no appreciable air resistance. When it has reached a height of 600 m , its...
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
The Dow process for synthesizing phenol, which involves the reaction of chlorobenzene with aqueous sodium hydroxide at high temperature and pressure, produces both diphenyl ether (1) and...
-
What are compounds A and B? Compound B has a strong IR absorption band in the 16501730 cm 1 region and a broad strong band in the 32003550 cm 1 region. 1-Methylcyclohexene (1) OSO (2) NaHSO A...
-
What risk arises if IT systems are not properly aligned to each other?
-
Preparation of a tax return for a partnership taxpayer Your client Nicole and Sam attended your office to have their 2 0 2 3 - year tax return prepared. Nicole Mania & Sam Baker are equal partners of...
-
5. An entity owned 100% of another entity's preference shares and 20% of ordinary shares. The investee's share capital outstanding at year-end included P5,000,000 of 10% cumulative preference shares...
-
How do prognostic methodologies leverage sophisticated algorithms and data analytics to generate nuanced forecasts across diverse domains, ranging from economics to climate science ?
-
To what extent does organizational culture, leadership ethos, and employee empowerment initiatives shape the fabric of operational efficiency within organizations, fostering a sense of ownership,...
-
Risk and Return Suppose you observe the following situation: Security Cooley Inc Moyer Company Beta Expected Return 1.8 22.00% 1.6 20.44 If the risk-free rate is 7%, are these securities correctly...
-
In each case, either prove the assertion or give an example showing that it is false. (a) P2 has a basis of polynomials f(x) such that f(b) = 0. (b) Every basis of M22 contains a noninvertible...
-
Modify the counter from Exercise 5.44 such that the counter will either increment by 4 or load a new 32-bit value, D, on each clock edge, depending on a control signal Load. When Load = 1, the...
-
Show the products of thesereactions: H2 b) a) Pd Pt 2 . IH2 c) CH;C=CCH,CH; d) CH;C=CCH,CH3 Lindlar catalyst Pd 2 H2 Pt
-
Show all of the steps in the mechanism for this reaction and explain the regiochemistry of theaddition: Br Br + HBr
-
Show the products of thesereactions: HCI . H,SO, b) a)
-
The wheels of a skateboard roll without slipping as it accelerates at 0.45 m/s down an 80-m-long hill. Part A If the skateboarder travels at 1.5 m/s at the top of the hill, what is the average...
-
A 2.30 g air-inflated balloon is given an excess negative charge, q = -3.50 10-8 C, by rubbing it with a blanket. It is found that a charged rod can be held above the balloon at a distance of d =...
-
A positive charge q = 2.60 C on a frictionless horizontal surface is attached to a spring of force constant k as in the figure shown below. When a charge of 42 = -8.350 C is placed 9.50 cm away from...

Study smarter with the SolutionInn App


