Birch reduction of toluene leads to a product with the molecular formula C 7 H 10 .
Question:
Birch reduction of toluene leads to a product with the molecular formula C7H10. On ozonolysis followed by reduction with dimethyl sulfide, the product is transformed into
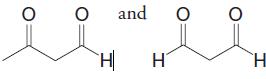
What is the structure of the Brich reduction product?
Transcribed Image Text:
and H H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
ans 1 Methy...View the full answer

Answered By

KATTUBOINA KOTA SATYA SAIKUMAR
EDUCATION;
I qualified CSIR-NET EXAM in december 2019.i got 49th rank in LS category,and also i cleared APSET exam which is conduct by ANDHRA UNIVERSITY in 2018.
M.SC (chemistry) from ANDHRA UNIVERSITY 2016-2018.
B.SC (MPC) from ADIKAVINANNAYYA UNIVERSITY 2013-2016.
Now i am working as chemistry lecturer in s.m.v.m.polytechnic college .
i can teach chemistry in iit-jee level and also i solve chemistry problems as simple as possible and give tricks to students for solve problems.i also help to students for enhancing their chemistry subject.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Question Posted:
Students also viewed these Sciences questions
-
There are three constitutional isomers with the molecular formula C 5 H 12 . Chlorination of one of these isomers yields only one product. Identify the isomer, and draw the product of chlorination.
-
Sabinene and 3-carene are isomeric natural products with the molecular formula C10H16. (a) Ozonolysis of sabinene followed by hydrolysis in the presence of zinc gives compound A. What is the...
-
Sabinene and 3-carene are isomeric natural products with the molecular formula C10H16. (a) Ozonolysis of sabinene followed by hydrolysis in the presence of zinc gives compound A. What is the...
-
As discussed throughout Chapter 3, the owner of Evergreen Solar (Jennifer) has been exploring different ways of performing predictive analytics in order to better predict whether any new sales lead...
-
The diprotic acid H2A has pK1 = 4.00 and pK2 = 8.00. (a) At what pH is [H2A] = [HA-]? (b) At what pH is [HA-] = [A2-]? (c) Which is the principal species at pH 2.00: H2A, HA-, or A2-? (d) Which is...
-
Fill in the blanks or answer true or false. -5s ||
-
Explain the significance of collection class and provide two examples.
-
The following selected financial data pertain to four unidentified companies: This financial information pertains to the following companies: a. Cable TV company b. Grocery store c. Accounting firm...
-
How do ethical leaders reconcile competing stakeholder interests while upholding principles of corporate social responsibility and integrity in decision-making ? Explain
-
MARK Partners is considering buying a service-oriented business at the end of 2019. Selected income statement and balance sheet items are provided below. MARK Partners expects revenue to grow...
-
Compound E has the spectral features given below. What is its structure? MS (m/z): M 202 IR (cm -1 ): 30303080, 2150 (very weak), 1600, 1490, 760, and 690 1 H NMR (): narrow multiplet centered at...
-
The reactions of aldehydes and ketones with LiAlH 4 and NaBH 4 (Section 12.3) are nucleophilic additions to the carbonyl group. What is the nucleophile in these reactions?
-
How would the volume of 1.00 cm3 of aluminum on Earth change if it were placed in a vacuum chamber and the pressure changed to that of the Moon (less than 109 Pa)?
-
What kinds of goals work best to build a team?
-
What is the most appropriate size for a decision-making group?
-
An automobile plant in a small town is rescued by Japanese ownership and an imported management team. Analyze this for teamwork and team building in the context of cultural differences. How do...
-
Is brainstorming a more effective creative process than nominal group technique?
-
Average but intriguing story about the human race being infected with small parasitic aliens called souls. Analyze from a systems perspective the isolated cave-dwelling humans who have created a...
-
Liu Corporation had the following transactions in debt instruments purchased to earn interest during the year ended December 31, 2014: Jan. 1 Purchased a 180-day (six-month) Canadian government...
-
What is the shape of the exponential distribution?
-
(a) Draw the resonance forms for SO2 (bonded O-S-O) (b) Draw the resonance forms for ozone (bonded O-O-O) (c) Sulfur dioxide has one more resonance form than ozone. Explain why this structure is not...
-
There is a small portion of the periodic table that you must know to do organic chemistry. Construct this part from memory, using the following steps. (a) From memory, make a list of the elements in...
-
For each compound, state whether its bonding is covalent, ionic, or a mixture of covalent and ionic. (a) NaCl (b) NaOH (c) CH3Li (d) CH2CI2 (e) NaOCH3 (f) HCO2Na (g) CF4
-
If you were running a campaign that had lower than expected impressions, what should you do?
-
1. Develop a definition for the Triple C model of project management. 2. List some of the factors that can impede the flow of information for project planning purposes. How can these factors be...
-
The Meat Mart has $900,000 in net income. The firm has 200,000 shares of stock outstanding. The market price per share is $76. What is the PE (price to earnings) ratio?

Study smarter with the SolutionInn App


