Compare the mechanisms of the following reactions: NH2 + NH2 + CI `N CI NH, NH, +
Question:
Compare the mechanisms of the following reactions:
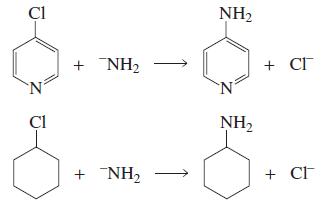
Transcribed Image Text:
NH2 + NH2 + CI `N CI NH, NH, + CF
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
here you have to understand mechanism of 1st reaction In 1st reaction 4chloropyridine treated with N...View the full answer

Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose mechanisms for the following reactions. (a) (b) (c) (d) heat cyclopentanol cyclopentene K. OH H SO4 heat pent-2-ene (cis trans) pentan-2-ol pent-1-ene H-SO heat CH3 CH2 CH CH3...
-
Propose mechanisms for the following reactions. (a) (b) (c) (d) DEH H PO heat CH,OH CH2 CH3 H,SO4 heat H2SO4 heat OH H,SOA heat
-
Propose mechanisms for the following reactions. (a) (b) (c) (d) CHPh OCH3 Ph + Ph COCH OCH OCH OH ) MVK (2) Ho N:
-
Find the mass and center of mass of the lamina that occupies the region D and has the given density function p. D is the triangular region with vertices (0, 0), (2, 1), (0, 3); (x, y) = x + y
-
Gold nanoparticles (Figure 16-29) can be titrated with the oxidizing agent TCNQ in the presence of excess of Br to oxidize Au(0) to AuBr in deaerated toluene. Gold atoms in the interior of the...
-
Determine the moment of inertia I x of the shaded area about the x axis. |-100 mm--100 mm150 mm 150 mm 150 mm 75 mm
-
What is the difference between null and alternative hypothesis?
-
January 1, 2017, Heckert Company purchases a controlling interest in Aker Company. The following information is available: a. Heckert Company purchases 1,600 shares of Aker Company outstanding stock...
-
The unadjusted trial balance for Ace Construction Company at its June 30 current fiscal year-end, is found on the trial balance tab. The credit balance of the V. Ace, Capital account was $53,660 on...
-
A propane-tired water heater delivers 80% of the standard heat of combustion of the propane [at 25(C with CO2(g) and H2O(g) as products) to the water. If the price of propane is $2.20 per gallon as...
-
Which measurement method would be most appropriate for the following items: historical cost, fair value, lower of cost and net realizable value, net realizable value, or present value? 1. Inventory...
-
Which measurement method would be most appropriate for the following items: historical cost, fair value, lower of cost and net realizable value, net realizable value, or present value? 1. Inventory...
-
Given the following account balances, determine the cash paid for insurance, office supplies, and wages in 2019. Prepaid insurance Office supplies Wages payable Insurance expense Office supplies...
-
What are the different types of advances of a bank according to RBI prudential norms?
-
You are the auditors of Apollo Services Ltd. and you have recently been reading a report on a similar company that criticised the auditors for failing to comment on the existence of the material...
-
What do you mean by Auditors Report?
-
Write short notes on compulsory tax audit.
-
What special points the auditor has to consider in conducting audit in the following institutions: (a) Charitable institution. (b) Educational institution.
-
How is the net present value Decision Rule related to cost-benefit analysis?
-
Rosalie owns 50% of the outstanding stock of Salmon Corporation. In a qualifying stock redemption, Salmon distributes $80,000 to Rosalie in exchange for one-half of her shares, which have a basis of...
-
Dimethyl sulfoxide (DMSO) has been used as an anti-inflammatory rub for race horses. DMSO and acetone appear to have similar structures, but the C=O carbon atom in acetone is planar, while the S=O...
-
Many naturally occurring compounds contain more than one functional group. Identify the functional groups in the following compounds: (a) Penicillin G is a naturally occurring antibiotic. (b)...
-
1. Draw a Lewis structure for each compound. 2. Label the hybridization, geometry, and bond angles around each atom other than hydrogen. 3. Draw a three-dimensional representation (using wedges and...
-
Determine the material inventory balance at the end of may? Received Issued Receiving Received Materials Report Number Received Quantity Unit Price Requisition Number Issued Quantity Issued Balance...
-
During October 2 0 2 3 , Fern Field Farms, Inc. received $ 1 0 , 0 0 0 from customers in exchange for fruit and vegetables. During the same month, the company paid $ 2 , 0 0 0 to employees, $ 5 0 0...
-
Given data below answer the question. Cash Accounts receivable $ 10,200 Cash dividends 15,200 Consulting revenue Office supplies 3,550 Rent expense $ 2,340 15,200 3,910 Office equipment 18,310 Land...

Study smarter with the SolutionInn App


