Question: For each molecule shown below, 1. indicate the most acidic hydrogens. 2. draw the important resonance contributors of the anion that results from removal of
For each molecule shown below,
1. indicate the most acidic hydrogens.
2. draw the important resonance contributors of the anion that results from removal of the most acidic hydrogen.
a.
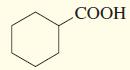
b.
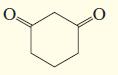
c.
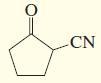
d.
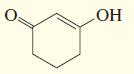
e.
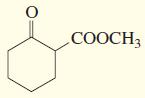
f.
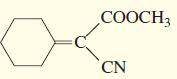
g.
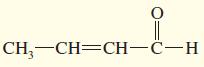
h.
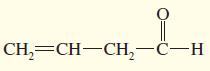
Step by Step Solution
3.41 Rating (151 Votes )
There are 3 Steps involved in it
A The given compound is cyclohexanoic acidThe most acidic hydrogen in that compound is carboxylic group of hydrogen due to the hydrogen is attached to ... View full answer

Get step-by-step solutions from verified subject matter experts


