Is the bicarbonate anion (HCO 3 ) a strong enough base to react with methanol (CH 3
Question:
Is the bicarbonate anion (HCO3) a strong enough base to react with methanol (CH3OH)? In other words, does the following reaction take place s written? (The pKa of methanol is 15.5; the pKa of H2CO3 is 6.4.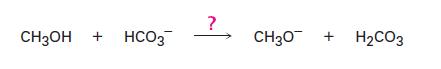
Transcribed Image Text:
СН3ОН + HCO3 ? CH3O + H2CO3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To determine if the bicarbonate anion HCO3 can act as a base and react with methanol CH3OH we need t...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
|1 5| = |5 m| a. Quantity A is greater. b. Quantity B is greater. c. The two quantities are equal. d. The relationship cannot be determined from the information given. Quantity A m Quantity B 4
-
Is the bicarbonate anion (HCO 3 - ) a strong enough base to react with methanol (CH 3 OH)? In other words, does the following reaction take place as written? CH3OH + HCO3 ? CH3O + HCO3
-
Is tert-butoxide anion a strong enough base to react with water? In other words, can a solution of potassium tert-butoxide be prepared in water? The pKa of tert-butyl alcohol is approximately18. CH...
-
What is path dependence? Explain the concept using your own words and provide an example of path dependence (other than what we have already mentioned in class). List the 4 categories of innovation,...
-
The account balances for Allied Electrical Supply, Inc., for the year ended August 31, 2012, are presented next in random order: Requirements 1. Prepare Allied Electrical Supplys single-step income...
-
In Problem 14, suppose that Douglas McDonnell shareholders approve a 3-for-1 stock split on January 1, 2017. What is the new divisor for the index? Calculate the rate of return on the index for the...
-
Use information from Section 6.7 to estimate which form of electromagnetic radiation is the lowest energy ionizing radiation. Data from section 6.7 When we first introduced the concept of the...
-
Crystal City established a capital projects fund to account for the construction of a new bridge. During the year the fund was established, the city issued bonds, signed (and encumbered) $6 million...
-
A beam of light travels from a vacuum into water at an angle of 45. The light has a frequency of 6.00 x 1014 Hz and travels at a speed of 2.26 x 108 m/s in water. The speed of light in a vacuum is...
-
Draw trans-1,2-dichlorocyclohexane in chair conformation, and explain why both chlorines must be axial or both equatorial.
-
The General Social Survey asked a random sample of 1,390 Americans the following question: "On the whole, do you think it should or should not be the government's responsibility to promote equality...
-
Which of the following statements is true? (a) Depreciation and amortization represent the cost of assets other than land that will benefit a business enterprise for less than a year. (b) Repair and...
-
Larsen & Toubro Ltd (www.larsentoubro.com) is an Indian multinational conglomerate listed on the Bombay Stock Exchange. In the financial year 2011, the companys net income was 4,456 crore, EPS was...
-
The rates of returns data from year 2000 to 2009 in Table 4.15 is listed below: At the = 0.05, can we conclude that the two stocks and the general market generate about the same average rate of...
-
A chicken farm came up with 4 different ways of mixing chicken feeds. The feeds were tested on 20 chickens. The results, given in terms of the chickens weight, are presented in the accompanying...
-
For these problems, use any web service that provides financial information. Good examples are Yahoo! Finance, Hemscott, Reuters and FT.com. You can also go to the companys website and download...
-
The performances of 250 salespeople in a company are summarized in the following table. Derive the expected frequencies, assuming that the data are from a normal distribution. Do the data collected...
-
You are considering purchasing some Stanley Ltd. common shares. Although Stanley recently had to incur significant expenditures for new capital assets, the company has been relatively profitable over...
-
Copy and complete the statement. 3800 m ? km =
-
(A true story.) While organizing the undergraduate stockroom, a new chemistry professor found a half-gallon jug containing a cloudy liquid (bp 100105 C), marked only STUDENT PREP. She ran a quick...
-
A (C-D) (carbondeuterium) bond is electronically much like a C-H bond, and it has a similar stiffness, measured by the spring constant, k. The deuterium atom has twice the mass (m) of a hydrogen...
-
The mass spectrum of n-octane shows a prominent molecular ion peak (m z 114). There is also a large peak at m/z 57, but it is not the base peak. The mass spectrum of 3,4-dimethylhexane shows a...
-
2023 Tax Rate Schedules Individuals Schedule X-Single If taxable income is over: But not over: $ 0 $ 11,000 $ 11,000 $ 44,725 $ 44,725 $ 95,375 $ 95,375 $ 182,100 $231,250 $ 578,125 $ 182,100 $...
-
IL. C. Body Boards Inc. Budgeting Project As the head accountant for L. C. Body Boards Inc., you are responsible for preparing the company's master budget for the first quarter of 20XX. The...
-
Reflective writing: Question & answer essay format. Provide peace of media (image/ screenshots of a clip or a link to a clip) of your choice for each answer. answer that is a thorough analysis of...

Study smarter with the SolutionInn App


