Tell whether each of the following compounds has the E or the Z configuration: a. b. c.
Question:
Tell whether each of the following compounds has the E or the Z configuration:
a.
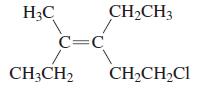
b.
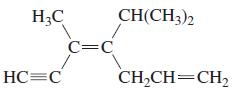
c.
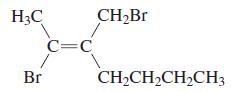
d.
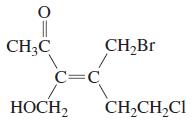
Transcribed Image Text:
H3C CH2CH3 C=C CH3CH2 CH2CH2CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
The E Z configurations can be identified by observation of the compounds attache...View the full answer

Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine whether each of the following compounds is a cis isomer or a trans isomer a. b. c. d. e. f. Cl Br CH3 CH3 Br Br CH3 Cl CH3 CH3 CH
-
Tell whether each of the following molecules has a meso stereoisomer. (a) (b) CH CHCH CHCH Cl CH, CHCH2CH,CH CI
-
Identify whether each of the following compounds is chiral or achiral: a. b. c. d. e. f. g. h. i. j. k. l. m. n. o. p. CI
-
Charles has a savings account with a balance, today, of 100,000 SAR in his investment account. He expects to obtain a yearly return of 12% in his investments. How long will it take for Charles to...
-
(a) The mean free path is the average distance a molecule travels before colliding with another molecule. The mean free path,, is given = KTI (2P) by where k is Boltzmann's constant, T is...
-
The tower crane is used to hoist a 2-Mg load upward at constant velocity. The 1.5-Mg jib BD and 0.5-Mg jib BC have centers of mass at G 1 and G 2 , respectively. Determine the required mass of the...
-
It has been reported that 30 percent of all couples who adopt a child because of troubles conceiving a child eventually do conceive a child after the adoption, suggesting that adoption makes...
-
See if you can determine the amount of Bluebird State Banks current net income after taxes from the figures below (stated in millions of dollars) and the amount of its retained earnings from current...
-
Zama CC acquired Machine A ( a second - hand machine ) for R 2 0 0 0 0 0 0 and brought it into use in its manufacturing process on 1 0 January 2 0 1 7 . Zama CC acquired a new manufacturing machine B...
-
Viscous heating in a ball-point pen, you are asked to decide whether the apparent decrease in viscosity in ball-point pen inks during writing results from "shear thinning" (decrease in viscosity...
-
The rate constant for a reaction can be increased by _____ the stability of the reactant or by _____ the stability of the transition state.
-
Which of the following carbocations would you expect to rearrange? a. b. CH2 CH3 CH3 e. CH;CHCHCH3 .
-
When coin 1 is flipped, it lands on heads with probability .4; when coin 2 is flipped, it lands on heads with probability .7. One of these coins is randomly chosen and flipped 10 times. (a) What is...
-
Why is the doctrine of joint and several liability criticized?
-
True Or False Under the rule of joint and several liability a defendant can be held responsible for all of the damages even though their contribution to the plaintiffs injuries was relatively minor.
-
True Or False Those who maintain that the tort system is too plaintiff-oriented point to the expansion of strict liability and the adoption of comparative negligence.
-
Joint tortfeasors are jointly and severally liable for any harm that is ____________.
-
How do the Progressive Era reformers and the classical reformers view joint and several liability? a. What are the conflicting objectives that dominate any discussion of joint and several liability?...
-
What is the reasoning behind using the average annual return as a measure of expected return?
-
Determine whether the lines are parallel, perpendicular, or neither. 2x + 3y = -12, 2y - 3x = 8
-
Tributylamine has a Cl mass spectrum with a strong M + 1 peak and one other major peak resulting from a-cleavage. At what m/z value does this peak occur?
-
In the warehouse of the company Tumany Amines, Iilc., two unidentified compounds have been found. The president of the company, Wotta Stench, has hired you to identify them from their spectra:...
-
In the warehouse of the company Tumany Amines, Iilc., two unidentified compounds have been found. The president of the company, Wotta Stench, has hired you to identify them from their spectra:...
-
For Kosko Company, actual sales are $1.218,000 and break-even sales are $962,220. Compute (a) the margin of safety in dollars and (b) the margin of safety ratio. Margin of safety Margin of safety...
-
Sheridan Corp. had total variable costs of $224.200 total hied costs of $143500, and total revenues of $380.000. Compute the required sales in dollars to break even
-
Consider the following information: Raw materials inventory, beginning $4,000 Raw materials inventory, ending 3,000. Net Purchases 25,000 Freight out 500% Decrease in Work in Process inventory Wip...

Study smarter with the SolutionInn App


