When fumarate reacts with D 2 O in the presence of the enzyme fumarase, only one isomer
Question:
When fumarate reacts with D2O in the presence of the enzyme fumarase, only one isomer of the product is formed. Its structure is shown. Is the enzyme catalyzing a syn or an anti addition of D2O?
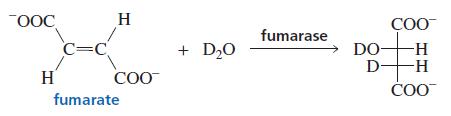
Transcribed Image Text:
OOC H COO fumarase C=C + D20 DO- D- H H- H COO COO fumarate
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
The addition of D 2 O across the double bond can occur in either s...View the full answer

Answered By

Sachin Nalawade
EDUCATION:
PhD. (SRF) in Chemistry, Department of Chemistry
Indian Institute of Science Education and Research (IISER), Pune, India.
Master of Science (Chemistry)Department of Chemistry,
Dr. Babasaheb Ambedkar Marathwada University, Aurangabad.
Bachelor of Science (Chemistry, Physics, Comp.Sci.)
Rajarshi Shri Shahu Arts, Commerce and Science College, Pathri.
Teaching Experience :
Served as teaching assistant during doctoral program for three different undergraduate courses in chemistry (including one practical course) and problem solving sessions at Indian Institute of Science Education and Research (IISER), Pune, India.
I have solid experience in research and great scientific knowledge in chemistry, great problem
solving ability, adaptability to change, ability to work in team, good in communication
and social awareness, experience in research for more than five year and teaching for 2
year.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) When fumarate reacts with D2O in the presence of the enzyme fumarase only one stereoisomer of deuterated malate is formed, as shown in Fig. P7 .52.Is this a syn- or an ant I - addition ? Explain....
-
When toluene reacts with chlorine gas in the presence of an iron(III) catalyst, the product is a mixture of the ortho and para isomers of C6H4ICH3. However, when the reaction is light-catalyzed with...
-
When the hydration of fumarate is catalyzed by the enzyme fumarase in D2O, only (2s,3R) 3-deuteriomalate is formed as the product. (Each -CO, group is the conjugate base of a carboxylic acid group.)...
-
What is the process of closing the space between a particular ability and the desired and necessary skill and achieving a more well-rounded and balanced CVF profile?
-
A solution was prepared by mixing 10.00 mL of unknown (X) with 5.00 mL of standard (S) containing 8.24 g S/mL and diluting the mixture to 50.0 mL. The measured signal quotient was (signal due to...
-
Beam AB has a negligible mass and thickness, and supports the 200-kg uniform block. It is pinned at A and rests on the top of a post, having a mass of 20 kg and negligible thickness. Determine the...
-
What are the advantages and disadvantages of judgmental sampling?
-
1. What aspects of U.S. and Indian culture may have been causes of Cokes difficulties in India? 2. How might Coca-Cola have responded differently when this situation first occurred, especially in...
-
Solve the following recurrence relations. You may use any method discussed in class, but you need to justify your answers by showing all relevant details. If you use a guess-and-prove method,...
-
Slicker is a growing, monocentric city surrounded by 500 miles of farmland in all directions. Land Use Industrial Commercial Residential Amount of Rent Earned Per Acre/Year/Land-Use Type at the...
-
Sherry O. Eismer decided that the configuration of the asymmetric carbons in a sugar such as D glucose could be determined rapidly by simply assigning the R configuration to an asymmetric carbon with...
-
A chemist wants to synthesize 4-decyne but cannot find any 1-pentyne, the starting material used in the synthesis just described. How else can 4-decyne be synthesized?
-
Why is there no need for CSMA/CD on a full-duplex Ethernet LAN?
-
How can the Internet be used to support the different stages of the buying process?
-
Which techniques can be used to increase awareness of a brand and encourage interaction with the brand?
-
Discuss the extent to which it is possible to operate as a virtual organisation.
-
Explain the purpose of environmental scanning in an e-commerce context.
-
Internet access varies from country to country; explain the key factors which might influence different levels of access.
-
If depreciation expense is not a cash flow, why do we have to subtract it and add it back? Why not just ignore it?
-
Refer to Exercise 8.S.I. Construct a scatterplot of the data. Does the appearance of the scatterplot indicate that the pairing was effective? Explain. Exercise 8.S.I. A volunteer working at an animal...
-
The stability of a DNA double helix can be measured by its melting temperature, Tm, defined as the temperature at which the helix is 50% dissociated into individual chains. Which of the following...
-
When DNA is treated with 0.5 M NaOH at 25C, no reaction takes place, but when RNA is subjected to the same conditions, it is rapidly cleaved into mononucleotide 2- and 3-phosphates. Explain.
-
Indicate whether you would expect imidazole to have high or low water solubility, and why.
-
If you were to enter the banking industry, you might find yourself approving or not approving loans. The following is a good example of a common event you could encounter. Company X is looking for...
-
You see a two year bond with annual coupon rate of 8% that has these flows. Years ahead Cashflows 0 ?? 0.25 0 0.5 0 0.75 1 1.25 1.5 1.75 2 8 0 0 0 108 You will be asked to calculate its price at...
-
Use synthetic division and the Remainder Theorem to evaluate P(c). P(x) = 6x + 5x +9, c = 11/1 P(1) =

Study smarter with the SolutionInn App


