Which amino acids in Table 23.1 have more than one asymmetric carbon? Table 23.1 The Most Common
Question:
Which amino acids in Table 23.1 have more than one asymmetric carbon?
Table 23.1
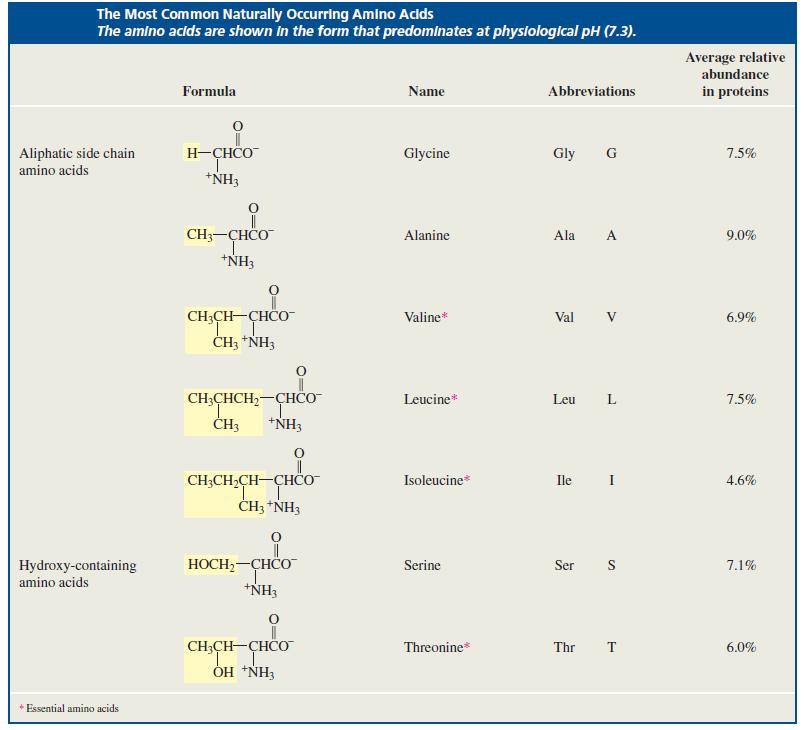
Transcribed Image Text:
The Most Common Naturally Occurring Amino Acids The amino acids are shown in the form that predominates at physiological pH (7.3). Average relative abundance Formula Name Abbreviations in proteins Aliphatic side chain H-ÇHCO Glycine Gly G 7.5% amino acids *NH3 CH3-CHCO Alanine Ala A 9.0% *NH3 CH;CH-CHCO Valine* Val V 6.9% ČH3 *NH3 CH;CHCH,-CHÖ *NH3 Leucine* Leu L 7.5% ČH3 CH;CH,CH-CHCO Isoleucine* Ile I 4.6% ČH3 *NH3 HOCH2-CHCO 7.1% Hydroxy-containing amino acids Serine Ser S *NH3 CH;CH-CHCO 1. ÓH *NH3 Threonine* Thr T 6.0% + Essential amino acids
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
Asymmetrys Carbon It is also called chiral carbon It is defined as that carbon ...View the full answer

Answered By

Shantanu Jana
I have completed my schooling from contai model institution....after that I completed my graduation from vidyasagar university on chemistry....after that now I am studying in jadavpur university on chemistry.....I am going through teaching for many years since my graduation....so I hope I will be able to taught them.... I just won't be there teacher there I will also guide them to be a better human besides being a brilliant student....I hope they will cooperate with me....
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the amino acids in Table 27.1 have more than one stereogenic center? Table 27.1 TABLE 27.1 -Amino Acids Found in Proteins (Continued) Abbreviation Structural formula* Name Amino acids with...
-
Which of the amino acids in Table 27.1 have more than one stereogenic center?
-
Which amino acids in the urea cycle are the links to the citric acid cycle? Show how these links occur
-
Refer to the situation described in P 192. Assume Pastner measures the fair value of all options on January 1, 2021, to be $4.50 per option using a single weighted-average expected life of the...
-
What is a Clark electrode, and how does it work?
-
An experiment was conducted on a chemical process that produces a polymer. The four factors studied were temperature (A), catalyst concentration (B), time (C), and pressure (D). Two responses,...
-
What are the types of internal recruiting?
-
The following information is available for Bernard Corporation: Net income ................. $206,000 Decrease in accounts receivable ......... 4,900 Increase in inventory ............ 15,300...
-
Reporting Issuance and Retirement of Long-Term Debt On the basis of the details of the following bonds payable and related discount accounts, indicate the items to be reported in the Financing...
-
White Ski Resorts operates a series of ski resorts in northern Europe and reports under IFRS. On June 30, 20X0, White purchased land for 3,000,000. White reports land values on the balance sheet...
-
Marcella Ltd. (ML) is a Northern Ontario based manufacturer of building materials. In the fourth quarter of 20X1, MLs board of directors agreed with senior management that the company needed to...
-
The following transactions have been encountered in practice. Assume that all amounts are material. a. A company decided to put the assets of one product line up for sale (intended to be sold within...
-
Using EDGAR (Electronic Data Gathering, Analysis, and Retrieval system), find the annual report (10-K) for Coca-Cola and Pepsico for the year ended December 2019. Locate the Consolidated Statements...
-
Why is word of mouth more powerful than an advertised message?
-
Mobile has a diverse array of advertising options. What are some of the most popular, and how can each help visually and verbally express the key consumer benefit?
-
What are the pros and cons of Internet and social media marketing?
-
Why is customer service considered a must for an IMC campaign?
-
Why are more brands incorporating alternative media into their promotional mix?
-
Given the U.S. Corporate Tax Rate Schedule in Exhibit 10.6, what is the marginal tax rate and average tax rate of a corporation that generates a taxable income of $12 million in 2012?
-
When you weigh yourself on good old terra firma (solid ground), your weight is 142 lb. In an elevator your apparent weight is 121 lb. What are the direction and magnitude of the elevator's...
-
Draw the structure that corresponds with each name. (a) 3-ethyloctane (b) 4-isopropyldecane (c) Sec-butylcycloheptane (d) 2,3-dimethyl-4-propylnonane (e) 2,2,4,4-tetramethylhexane (f)...
-
Each of the following descriptions applies to more than one alkane. In each case, draw and name two structures that match the description. (a) An isopropylheptane (b) A diethyldecane (c) A...
-
Give the IUPAC names of the following alkanes. (a) CH3C(CH3)2CH(CH)CH3)CH2CH2CH(CH3)2 (b) (c) (d) (e) (f) (g) (h) CH,CH CHCH CH, CH CH CH,CHCH CH,CHCH CH,CH CH,CH, CH, CH,CH, CH,CH,CH, C(CH,CH),...
-
The price of a car you want is $39,000 today. Its price is expected to increase by $1000 each year. You now have $23,500 in an investment account, which is earning 11% per year. How many years will...
-
Marshall's concept of external economies and diseconomies refers to: a) Changes in output resulting from changes in input levels b) The effects of production on the environment c) The benefits or...
-
The DD-AA model predicts that a permanent increase in foreign money demand will lead to an improvement in the domestic current account. True/False/Uncertain, explain and support your answer with ONE...

Study smarter with the SolutionInn App


