Which of the following alkyl halides is most likely to undergo substitution by an SN 1 mechanism?
Question:
Which of the following alkyl halides is most likely to undergo substitution by an SN1 mechanism?
a.
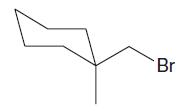
b.
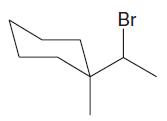
c.
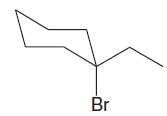
Transcribed Image Text:
Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
The most important factor is the structure of the carbon skeleton A helpful generalization is t...View the full answer

Answered By

Nakul Naskar
I am a organic chemist. I have experience of teaching organic chemistry to bring out Concepts about this subject which helps a student to enhance his/her knowledge and make a good impact on his educational life. Besides I can also teach Inorganic and physical chemistry.
I can monitor student performance or assist students in academic environments, such as classrooms, laboratories.
By Providing feedback to students using positive reinforcement techniques to encourage, motivate, or build confidence in students.
Prepare lesson plans or learning modules for tutoring sessions according to students' needs and goals.
During my teaching life I Maintain records of students' assessment results, progress, feedback, or school performance, ensuring confidentiality of all records.
I can clear your all doubts on chemistry by providing easy explanation.
Thank you
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Question Posted:
Students also viewed these Sciences questions
-
Which of the following two alkyl halides would react most rapidly in a solvolysis reaction by the SN1 mechanism? Explain your reasoning. Ch 1,0-CH-CH-CH,-Cl A (trans isomer) CH.O-C-CH,-CI CH2
-
Which of the alkyl halides is more reactive in an E2 reaction? a. CH3CH2CH2Br or b. c. d. CH,CH2CHCH3 Cl or CH cHy CH3CHCH2CHCH or CH3CH2CH2CCH3 Br Br CH, CH CH3 CH3
-
Which of the following alkyl halides form a substitution product in an SN1 reaction that is different from the substitution product formed in an SN2 reaction? a. b. c. d. e. f. CH Br CH CHCHCHCH CHa...
-
In Exercises, find the limit. x-4 lim x-00x + 1
-
Sketch the general appearance of the curve for the titration of a weak diprotic acid with NaOH. Explain (in words) what chemistry governs the pH in each distinct region of the curve.
-
Paul Monroe, CPA, has been in public practice and a member of the AICPA for 25 years. He signs over 300 individual tax returns each year, most of which have been initially prepared by his staff and...
-
What are the characteristics and advantages of the graphical user interface (GUI)?
-
1. Prepare an outline of the benefits that DH might expect to obtain from this CRM project. Use categories to organize your list of benefits; for example, you might identify benefits that will accrue...
-
What is "Leveraging Customer Knowledge"? Identify two methods employed by companies to improve the availability and use of customer knowledge. (5 Marks) Compare the strategic role of brands for...
-
Ming, CPA, is engaged to audit the financial statements of Wellington Sales, Inc., for the year ended December 31, 20X0. Ming obtained and documented an understanding of the clients business and...
-
Rank the following in terms of decreasing nucleophilicity in a protic solvent. CH 3 CO 2 - CH 3 O - CH 3 S - CH 3 SH CH 3 OH
-
Which would you expect to be the stronger nucleophile in a polar aprotic solvent? a. CH 3 CO 2 - or CH 3 O - b. H 2 O or H 2 S c. (CH 3 ) 3 P or (CH 3 ) 3 N
-
What are the stages in the knowledge acquisition process? Why is knowledge acquisition often called a bottleneck of the process of knowledge engineering? How can the acquired data affect our choice...
-
The following table sets out information in respect of Division X and Division Y. The cost of borrowing new finance is 10% per annum. Required Explain what view the managers of each division might...
-
As technology becomes more sophisticated and complicated, which challenges presented by virtual teams might become more problematic?
-
This extract describes changes in the UK company Bunzl, which announced at the start of 2005 that it would sell Filtrona, a business making cigarette filters, and concentrate on its outsourcing...
-
Explain what is meant by the accrual of liabilities.
-
Describe the main contents of a business plan for presenting to a bank providing finance for a small business.
-
Under the direct method, why is depreciation expense not reported in the operating activities section?
-
Decades after the event, Johnson & Johnson (J&J), the 130-year-old American multinational, is still praised for swiftly The company indicated that its response was based on the expectations set forth...
-
Suppose a mixture of AQC-amino acids is subjected to HPLC on a stationary phase that consists of C8-silica rather than Cl 8-silica; that is, the glass stationary phase (Eq. 26.34, p. 1293) contains...
-
Sometimes it is necessary in solid-phase peptide synthesis to use a resin linker that is more sensitive (that is, more reactive) to acid than the linker shown in Eq. 26.21 on p. 1285. The following...
-
Sometimes it is necessary in solid-phase peptide synthesis to use a resin linker that is more sensitive (that is, more reactive) to acid than the linker shown in Eq. 26.21 on p. 1285. The following...
-
Record the following transactions for Tri - State Corporation on the dates indicated. 1 . On March 3 1 , 2 0 2 0 , Tri - State Corporation discovered that Depreciation Expense on equipment for the...
-
William has decided to increase his sales price to $ 2 6 to offset the supplier's price increase. He believes that the increase will result in a 5 % reduction from last year's sales volume. What is...
-
The same scenarios exist as before, with labor costing either $ 1 3 / lawn or $ 2 5 / lawn . Assume that your mowing business will mow 3 5 0 lawns per month rather than 2 0 0 lawns per month. Also...

Study smarter with the SolutionInn App


